A review of the extensive clinical trial landscape shows how CAR T therapies are moving beyond single-target designs to multifunctional “living drugs” designed to improve safety, overcome tumor escape, and tackle difficult-to-treat cancers.
Review: Mapping the clinical landscape of multifunctional CAR T cells: targets, trends, and synergies
In a comprehensive review recently published in the magazine molecular therapyGerman researchers analyzed 1,801 registered clinical trials to map the evolution of “next generation” chimeric antigen receptor (CAR) T cell products.
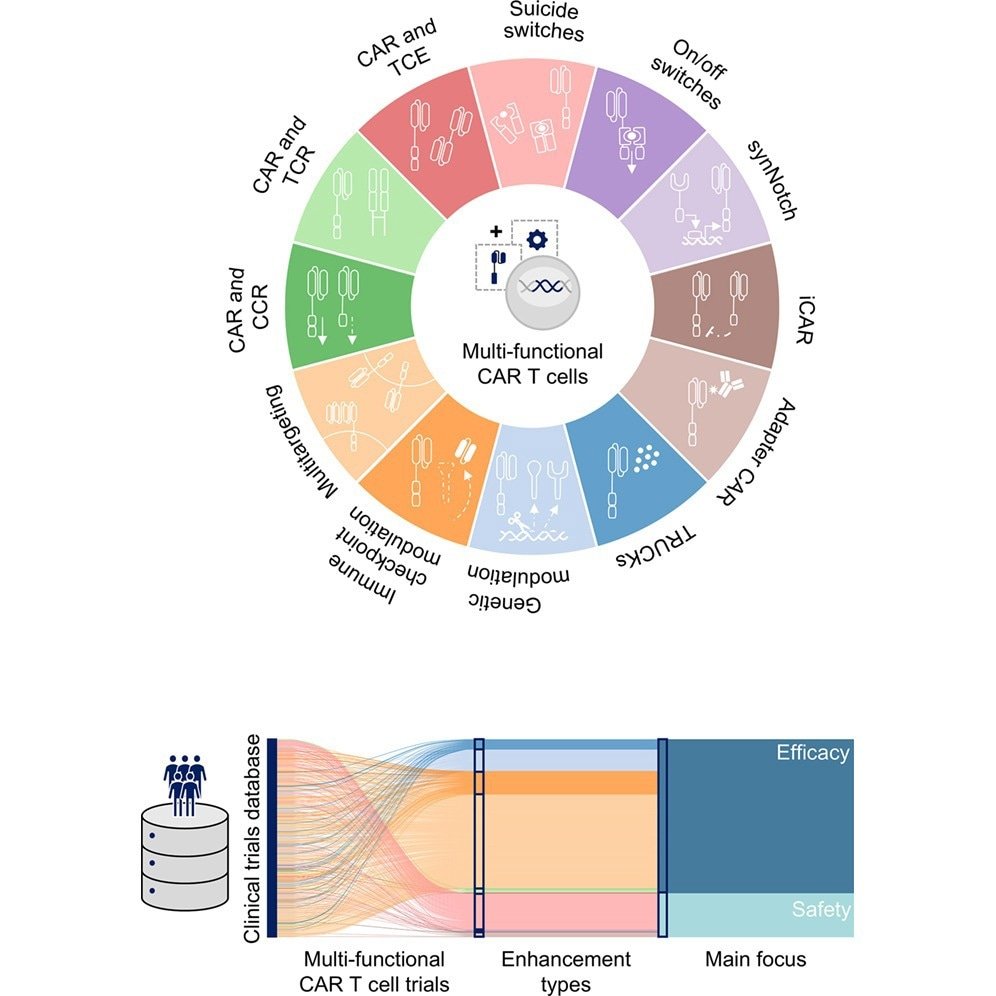
The review found that multifunctional CAR T-cell therapies accounted for 533 of 1,801 CAR T trials, representing 33% of new CAR T products submitted to clinical trials in 2025, and are rapidly moving from simple designs to complex multifunctional architectures that incorporate adjunctive features to enhance safety and efficacy, particularly in solid tumors, such as “suicide switches.”
This review concludes that while current efficacy-focused strategies such as multitargeting are growing rapidly and new combination designs are beginning to show promising potential in preclinical and early clinical settings, future impact will depend on overcoming manufacturing, regulatory, and clinical validation challenges.
Background of next-generation CAR-T cell therapy
Chimeric antigen receptor (CAR) T cells are a relatively new therapeutic approach that manipulates immune cells to recognize and destroy malignant tumors. The first of these “cell therapies” was approved for clinical use in humans by the U.S. Food and Drug Administration (FDA) in 2017. Since then, these treatments, colloquially referred to as “living medicines,” have been reported to have saved thousands of lives, especially in blood cancers, where they are most effective.
However, subsequent studies have revealed that the classic CAR T cell design has several significant drawbacks. 1. Some patients exhibited severe immune-mediated toxicities, including cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS), and 2. It had limited efficacy against solid tumors.
As a result, “classical” CAR designs are being complemented by research into next-generation “multifunctional” CAR T cells. These emergency measures are designed to incorporate safety enhancements such as “suicide switches” that can be activated externally if an adverse patient reaction is observed. Additionally, some CAR T cells are engineered to persist, recruit, or secrete cytokines that support antitumor activity, while others target multiple cancer markers simultaneously to prevent disease hiding.
Clinical trial review design and categories
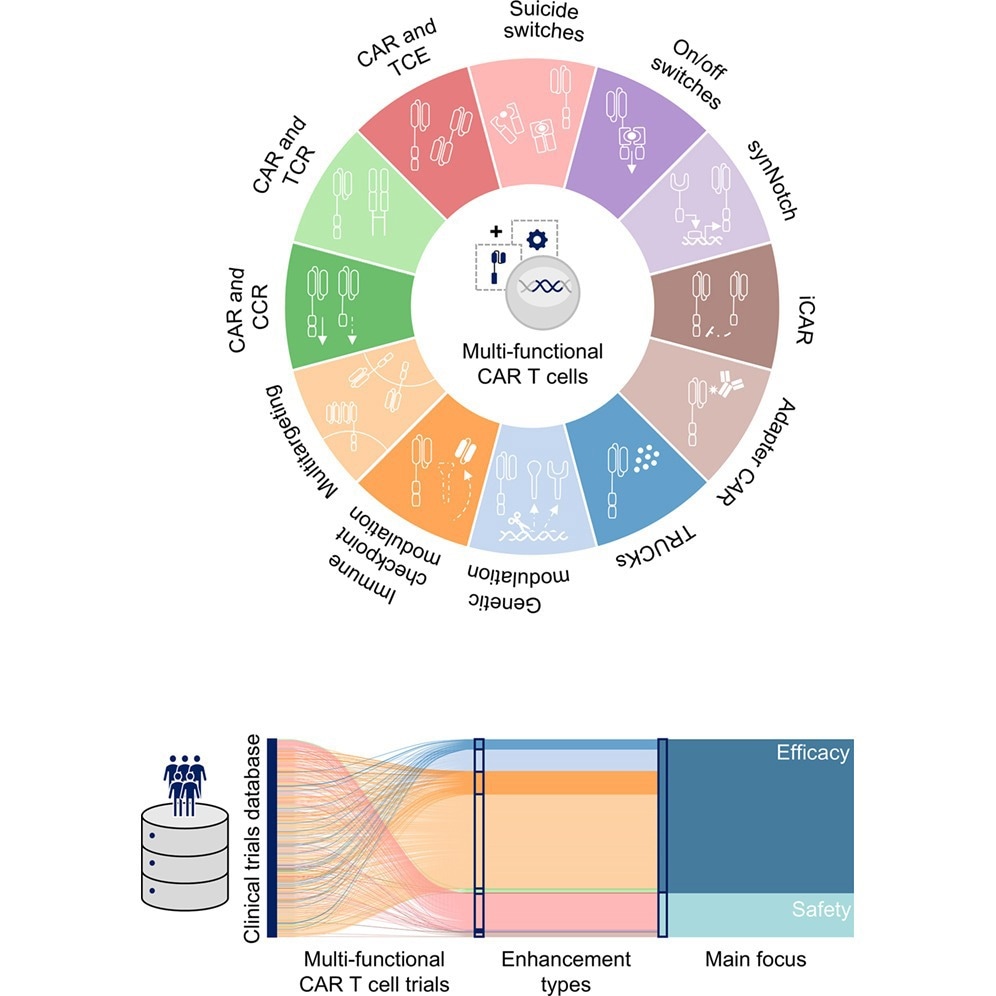
This review aims to synthesize the evolution of CAR T cells from their classical roots to new multifunctional variants through a data-driven approach. The review dataset was extracted and curated from relevant clinical trials (n = 1,801) registered on ClinicalTrials.gov between 2003 and September 2025.
The shortlisted publications were divided into two main categories:
Safety enhancement studies consist of technologies such as “on/off switches” or “suicide switches” (such as iCasp9), which allow physicians to monitor patient responses and inactivate or control CAR T cells if patients exhibit adverse side effects, and efficacy enhancement strategies, such as multitargeting (modalities that can identify and then target two or more distinct tumor antigens) and armored CARs (endogenously secreting cytokines that increase survival). and checkpoint regulation studies (designed to resist prolonged antigenic stimulation (“T-cell depletion”) and tumor microenvironment-induced immunosuppressive factors).
Statistical analyzes included the generation of UpSet plots (a relatively new data visualization approach for analyzing multidimensional sets and their intersections) and network analysis to map patterns, overlaps, and target combinations across different CAR T technologies (e.g., CAR in combination with T cell engager (TCE) or chimeric costimulatory receptor (CCR)).
Multifunctional CAR-T testing trends and findings
This review revealed that of the 1,801 studies analyzed, 533 (approximately 33%) investigated multifunctional products. Additionally, multi-target CAR T technologies accounted for 52% of multi-functional CAR T approaches in 2025.
Network analysis confirmed these findings and highlighted that while hematologic cancer targets (e.g., CD19, CD20, BCMA (B-cell maturation antigen)) remain the most common overall and hematologic indications still predominate in both mono- and multi-functional CAR T trials, urgent investigations are increasingly focused on acute myeloid leukemia (AML) and emerging clusters of solid tumors.
We found that these new studies, particularly those on solid tumors, are much more likely to incorporate combination strategies that exploit potency enhancers and suicide switches than their traditional CAR T counterparts.
After examining data from trials that incorporated suicide switches and similar safety mechanisms (more than 100), the review found that these switches rarely trip in clinical settings. However, the authors caution that more clinical evidence is needed before firm conclusions can be drawn about the switch’s relative safety and effectiveness, as the switch has only been activated in a small number of reported cases.
The review also notes the rapid expansion of CAR T trials targeting autoimmune diseases, increasing from 35 in 2023 to 185 in 2025, a more than five-fold jump in just two years, highlighting the movement of CAR T technology beyond oncology.
Future direction of multifunctional CAR-T cells
This review posits that the future of CAR T immunotherapy is likely to diverge from traditional “universal” cell designs to complex “trifunctional” cells. However, the authors highlight some key hurdles that need to be addressed before this future becomes a reality. 1. Difficulties in manufacturing these cells (and associated prohibitive costs), 2. Limited clinical outcome data for many approaches, and 3. Regulatory approval, including the need for a framework that can accommodate modular or composite CAR T designs.
Improving the efficacy of CAR T interventions in solid tumors and ensuring the safety of CAR T interventions in human patients will likely be the main focus of CAR T research efforts from now until 2030.
Click here to download your PDF copy.