Major depressive disorder (MDD) is a major global health problem and one of the leading causes of disability. Approximately 30% of people diagnosed with depression develop treatment-resistant depression (TRD). This means that standard antidepressants do not sufficiently improve symptoms. Ketamine has gained attention as a fast-acting antidepressant for TRD patients. However, scientists do not fully understand how this therapy works in the human brain, making it difficult to refine and personalize it.
New research published in molecular psychiatry On March 5, 2026, we set out to solve this mystery. The study was led by Professor Takuya Takahashi of the Department of Physiology, Yokohama City University Graduate School of Medicine, Japan. The research team used an advanced positron emission tomography (PET) imaging method to directly observe changes in the glutamate alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR). This receptor is an important protein that helps regulate communication between brain cells and plays a key role in synaptic plasticity and glutamatergic signaling in patients receiving ketamine.
Professor Takahashi explains, “Ketamine has shown rapid antidepressant effects in patients with treatment-resistant depression, but its molecular mechanism in the human brain remains unclear.”
Visualizing brain receptors using a new PET tracer
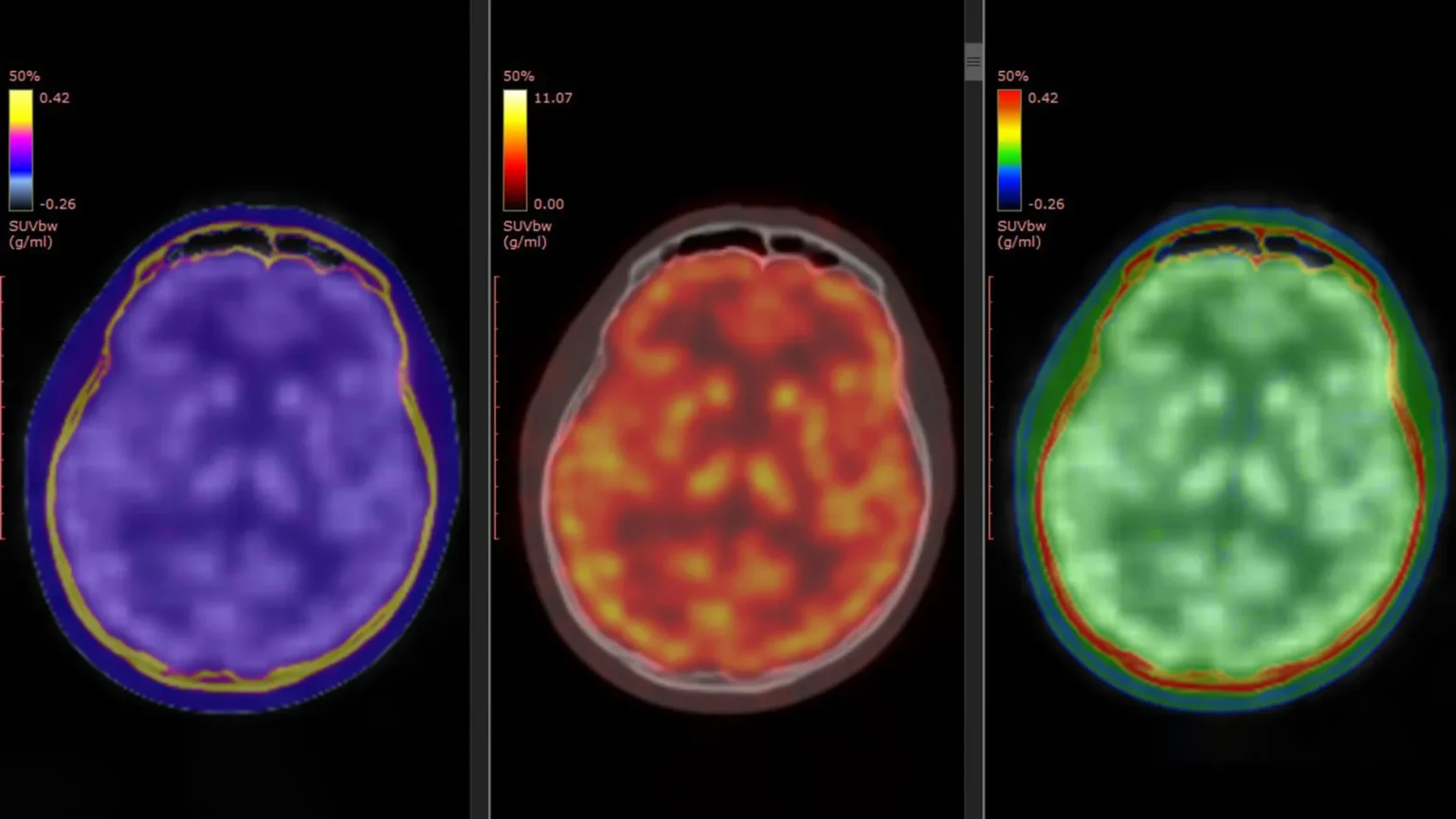
The study relied on a PET tracer previously developed by the team, known as (¹¹C)K-2. This tracer allows scientists to visualize cell surface AMPARs directly in the living human brain. Previous laboratory and animal studies suggest that ketamine’s antidepressant effects involve AMPAR activity. A new study provides the first direct evidence that this process is occurring in humans.
To conduct this study, researchers combined data from three registered clinical trials conducted in Japan. The study group included 34 patients diagnosed with TRD and 49 healthy participants as controls.
Patients received ketamine or a placebo intravenously for two weeks. PET brain imaging was performed before the start of treatment and again after the last injection. This approach allowed researchers to compare changes in AMPAR levels and distribution in the brain over time.
Region-specific brain changes associated with symptom relief
The results showed that TRD patients had a wide range of abnormalities in AMPAR concentrations compared to healthy subjects. These differences appeared in specific brain regions rather than throughout the brain.
Ketamine did not cause uniform changes throughout the brain. Instead, improvements in depressive symptoms were associated with dynamic region-specific adjustments in AMPAR levels. Although some cortical regions showed an increase in receptor density, regions associated with reward processing, particularly the habenula, showed a decrease. These region-specific changes were strongly associated with improvement in patients’ depressive symptoms.
“The antidepressant effects of ketamine in TRD patients are mediated by dynamic changes in AMPARs in the living human brain,” Professor Takahashi explained. “Using the new PET tracer (11C)K-2, we were able to visualize how ketamine changes AMPAR distribution across specific brain regions and how these changes correlate with improvement in depression symptoms.”
These observations support mechanisms previously identified in animal studies and provide direct human evidence linking them to actual clinical antidepressant effects.
Potential biomarkers for predicting treatment response
The results of this study do more than just reveal how ketamine works. They may also have practical clinical value. PET images of AMPARs may serve as a biomarker to help doctors assess and predict how TRD patients will respond to ketamine treatment.
Identifying reliable biological markers of treatment response remains an important goal in mental health care, as many patients do not respond to standard antidepressants.
Aiming for more individualized depression treatment
This study helps bridge the long-standing gap between laboratory research and clinical psychiatry by allowing scientists to directly observe AMPAR activity in living human brains. Our results identify AMPAR modulation as a central mechanism behind ketamine’s rapid antidepressant effects and suggest that AMPAR PET imaging may lead to more personalized treatment strategies in the future.
Ultimately, this research may help develop more precise treatments for people living with treatment-resistant depression.
This research was supported by the Ministry of Education, Culture, Sports, Science and Technology (Science and Technology Promotion Coordination Fund). Japan Agency for Medical Research and Development (AMED) (grant numbers: JP18dm0207023, JP19dm0207072, JP24wm0625304, JP25gm7010019, JP20dm0107124). Japan Society for the Promotion of Science Grants-in-Aid for Scientific Research (grant numbers: 22H03001, 20H00549, 20H05922, 23K10432, 19H03587, 20K20603, 22K15793, 21K07508) Takeda Science Foundation. Keio University Next Generation Research Project Program. Senshin Medical Research Foundation and Japan Clinical Pharmacology Research Foundation.