A comprehensive new review shows how gut microbiome disruption promotes obesity, insulin resistance, and cardiovascular risk, while pointing to dietary and other microbiome-focused strategies that may help restore metabolic balance.
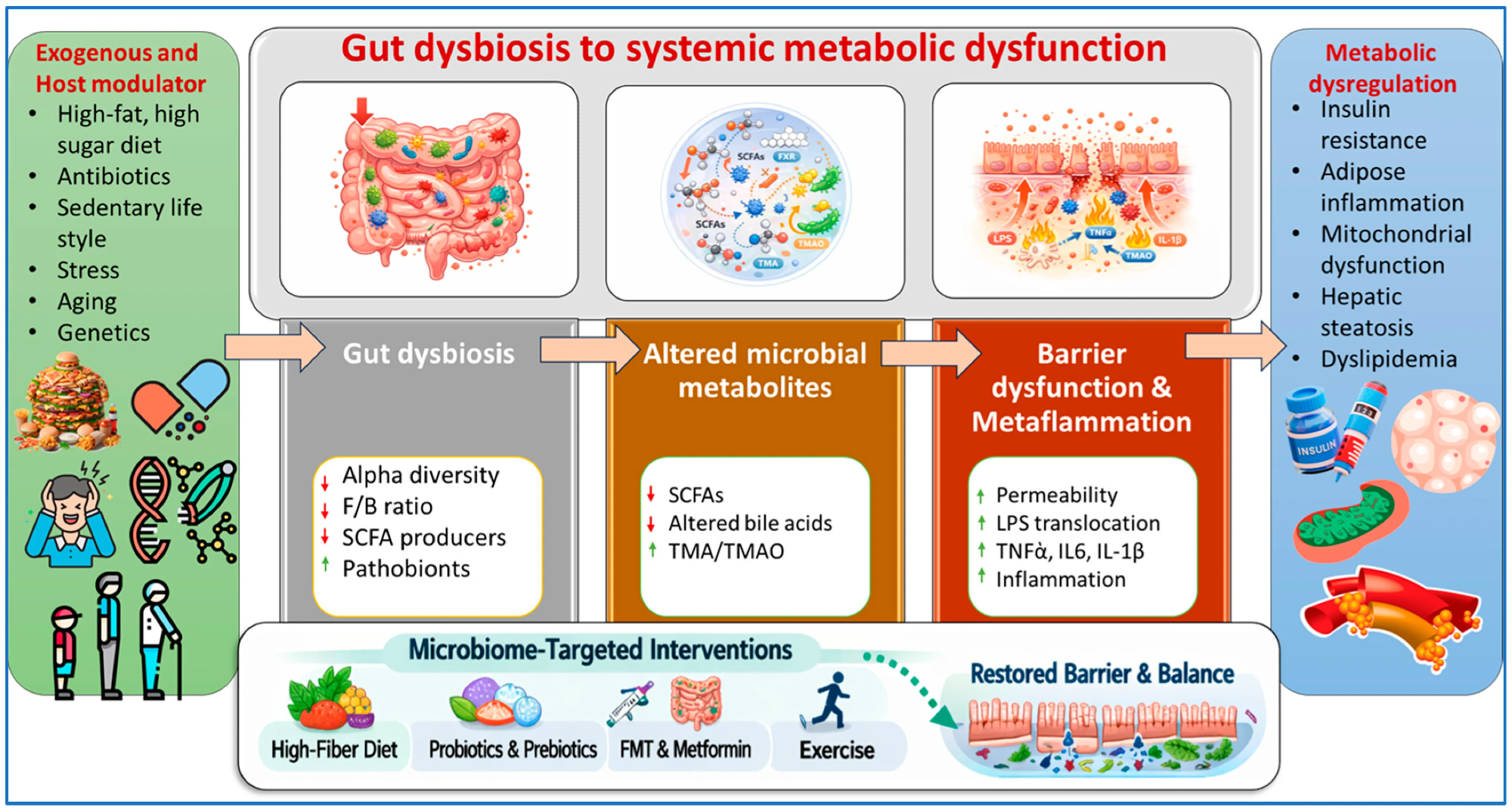
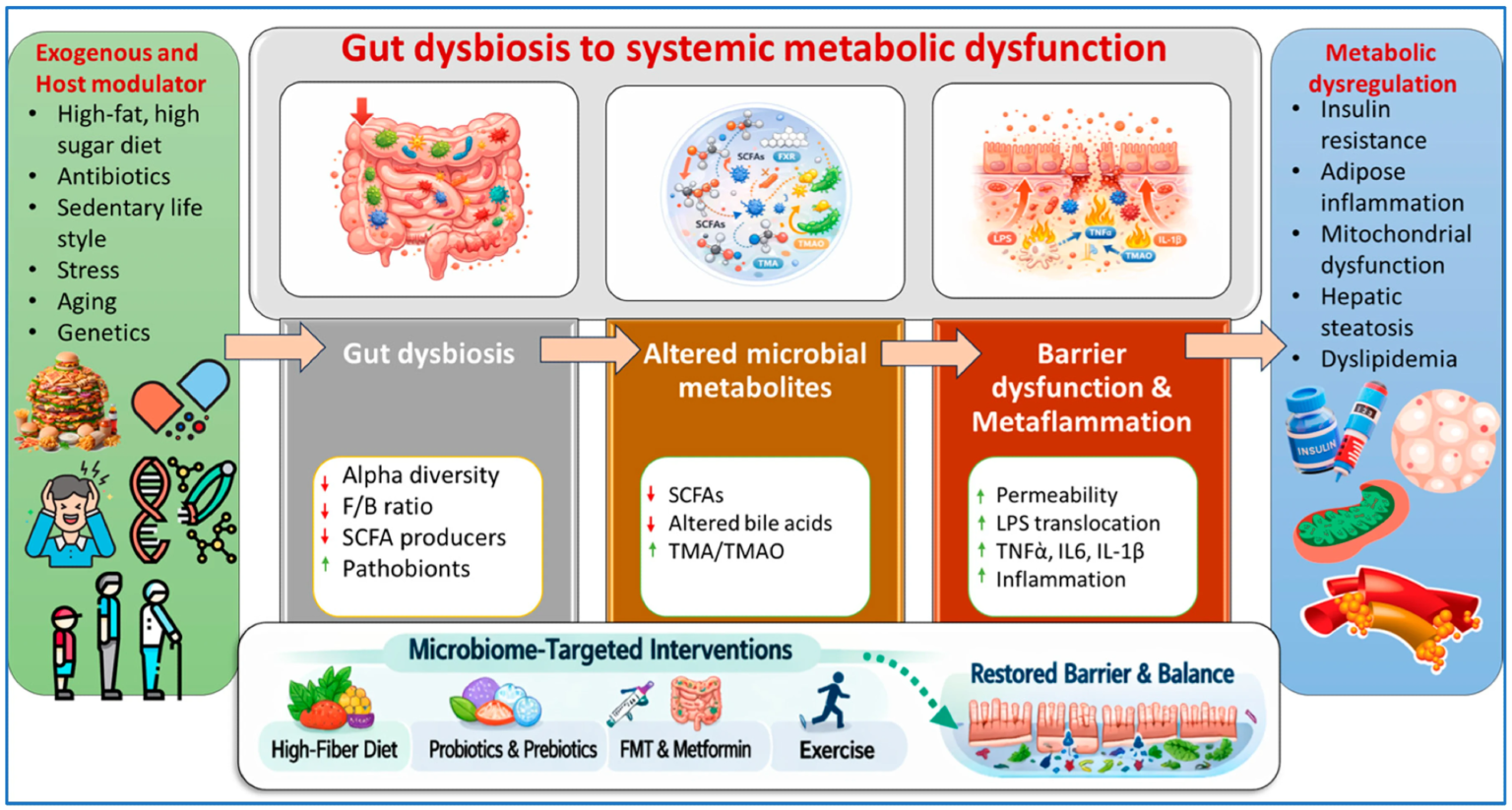
An integrative mechanistic framework linking dysbiosis to systemic metabolic dysfunction. Arrows indicate the progression of events from extrinsic and host-related factors to dysbiosis of the gut microbiota, changes in microbial metabolites, barrier dysfunction, and metabolic dysregulation. Upward (↑) and downward (↓) arrows indicate increase and decrease, respectively.
A growing body of evidence highlights the role of the gut microbiome in metabolic health. Recent reviews published in magazines nutrients We identify dysbiosis of the gut microbiome, a disruption of microbial composition and function, as a key factor associated with metabolic dysfunction through interrelated mechanisms, including oxidative stress, inflammation, and epigenetic changes. Disruption of intestinal barrier integrity allows toxins to migrate into the bloodstream and liver, where metabolic disorders can be amplified. These findings highlight the potential of gut-targeted strategies to improve cardiometabolic outcomes.
Gut microbes play a central role in host metabolism by converting dietary substrates into short-chain fatty acids (SCFAs) that support host and microbial functions while maintaining gastrointestinal homeostasis. However, most studies have isolated individual aspects of dysbiosis, such as microbial composition, specific mechanisms, or interventions such as fecal microbiota transplantation (FMT), rather than examining interrelated effects. Although the disorder is increasingly associated with metabolic disorders, the integrated pathways remain unclear. Future research should take a multidimensional approach that connects diet, exercise, genetics, therapeutics, and intestinal organ axes to better define a holistic model of metabolic health and overall well-being.
In this review, researchers analyzed the links between gut microbial imbalance, oxidative damage, immune pathways, epigenetic processes, and metabolic diseases. They focused on more recent literature, including both experimental and observational studies, and mechanistic findings from preclinical models, drawing on 161 peer-reviewed records in Scopus (2000 to January 2025).
Relationship between dysbiosis and systemic metabolic dysfunction
Dysbiosis contributes to cardiometabolic diseases through a network of metabolic, inflammatory, and barrier-related mechanisms. This is characterized by a decrease in microbial alpha diversity and a change in the Firmicutes/Bacteroidetes ratio, although this ratio alone is an oversimplified marker and may vary between populations and research methods. and other SCFA-producing bacteria are lost. rosebria seeds and Faecalibacterium prausnitziiand the spread of pathogens. Escherichia coli and Enterobacteriaceae. This review presents these taxa as context-dependent examples rather than universal biomarkers of dysbiosis.
Reduced availability of SCFAs, particularly butyrate, weakens epithelial tight junctions, impairs intestinal barrier function, and limits anti-inflammatory signaling. At the same time, increased intestinal permeability promotes the entry of lipopolysaccharide (LPS) into the circulation, activating Toll-like receptor (TLR)-mediated pathways and triggering the production of proinflammatory cytokines. These include tumor necrosis factor alpha (TNF-α), interleukin 1 beta (IL-1β), and interleukin 6 (IL-6), which collectively cause chronic low-grade inflammation and insulin resistance.
Microbial metabolites amplify metabolic risks. The intestinal bacterial community increases the production of trimethylamine (TMA), which is converted in the liver to trimethylamine N-oxide (TMAO), a pro-atherogenic molecule that promotes endothelial dysfunction and leukocyte activation. Changes in the microbiome also promote branched-chain amino acid synthesis. species such as prevotella cover and Bacteroides vulgatus Associated with insulin resistance. In obesity and type 2 diabetes, studies have reported reduced diversity, depletion of butyrate producers, and increase in opportunistic taxa. Clostridium citroniae.
These compositional and functional changes alter the bile acid pathway, intestinal hormone secretion such as glucagon-like peptide-1 (GLP-1) and peptide YY (PYY), and immune balance. The result is a self-reinforcing cycle of inflammation, metabolic dysregulation, and microbial imbalance that can help maintain obesity, insulin resistance, and cardiovascular disease. This review also points out that these relationships are often bidirectional, meaning that dysbiosis can act as both a cause and a consequence of metabolic dysfunction.
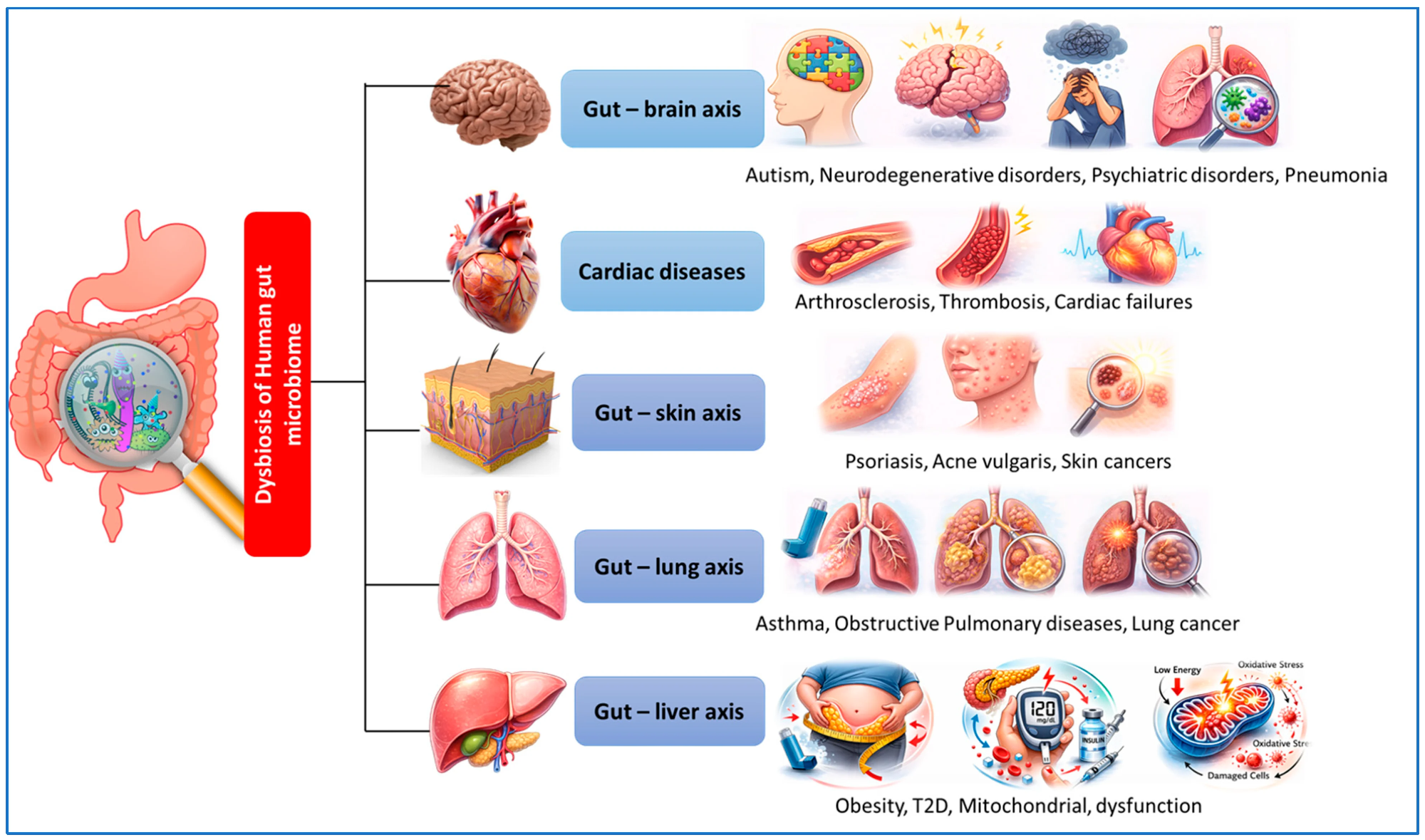
A conceptual overview of gut microbiota homeostasis and progression to gut microbiota-related metabolic dysfunction. The icons and schematic elements used in this diagram were created using AI-assisted graphics tools and were further modified and assembled by the author.
Interventions targeting the microbiome to improve metabolic health
Targeting the gut microbiome provides a practical way to improve cardiometabolic outcomes. Dietary strategies remain central. A high-fiber diet rich in fruits, vegetables, legumes, and whole grains promotes SCFA-producing microorganisms, increases epithelial barrier integrity, and improves insulin sensitivity. In contrast, a diet high in refined carbohydrates and saturated fat suppresses tight junction (TJ) protein expression, increases proinflammatory cytokines, alters bile acid signaling, and overall impairs intestinal barrier function. Probiotics and prebiotics further support microbial balance by promoting SCFA production, limiting pathogen overgrowth, and modulating immune responses. Emerging next-generation probiotics (NGPs) and postbiotics may offer more targeted metabolic benefits.
Lifestyle factors also shape microbial composition and function. Regular physical activity increases microbial diversity and butyrate production, and stress reduction practices such as mindfulness and meditation can help modulate gut-brain signaling and reduce inflammation. Sleep quality and circadian regulation further maintain microbial rhythms and metabolic balance. Conversely, irregular eating patterns, chronic stress, alcohol use, and medications such as antibiotics and proton pump inhibitors can cause dysbiosis and should be carefully managed.
FMT represents a more direct intervention. Clinical and experimental studies suggest that FMT, especially when combined with dietary interventions, may alter microbial composition, improve insulin sensitivity, and moderately alter fat distribution. Nevertheless, this review presents FMT as a promising approach rather than an established treatment for metabolic diseases.
Importantly, host factors such as genetics and aging also influence microbiome structure and metabolic responses, shaping individual differences in treatment outcomes. Epigenetic mechanisms, particularly deoxyribonucleic acid (DNA) methylation, further modulate this relationship, as microbial metabolites such as folate, S-adenosylmethionine, and SCFA can alter methylation patterns and gene expression. These changes affect metabolic and inflammatory pathways and contribute to disease susceptibility. Exploratory possibilities such as long-term epigenetic imprinting are also discussed in this review, but these are not yet well established.
Overall, the gut microbiota is an important regulator of metabolic and immune homeostasis, and internal metabolic abnormalities are associated with oxidative stress, inflammation, and epigenetic changes associated with cardiometabolic diseases. The evidence spans disparate studies of humans, animals, and machines, so further research is needed to disentangle causal relationships. Identifying early microbial markers and refining interventions are essential for targeted therapies. At the population level, policies promoting fiber-rich diets and limiting ultra-processed foods are important, while at the individual level, strategies focused on balanced nutrition, physical activity, stress management, and the microbiome may improve long-term health outcomes.