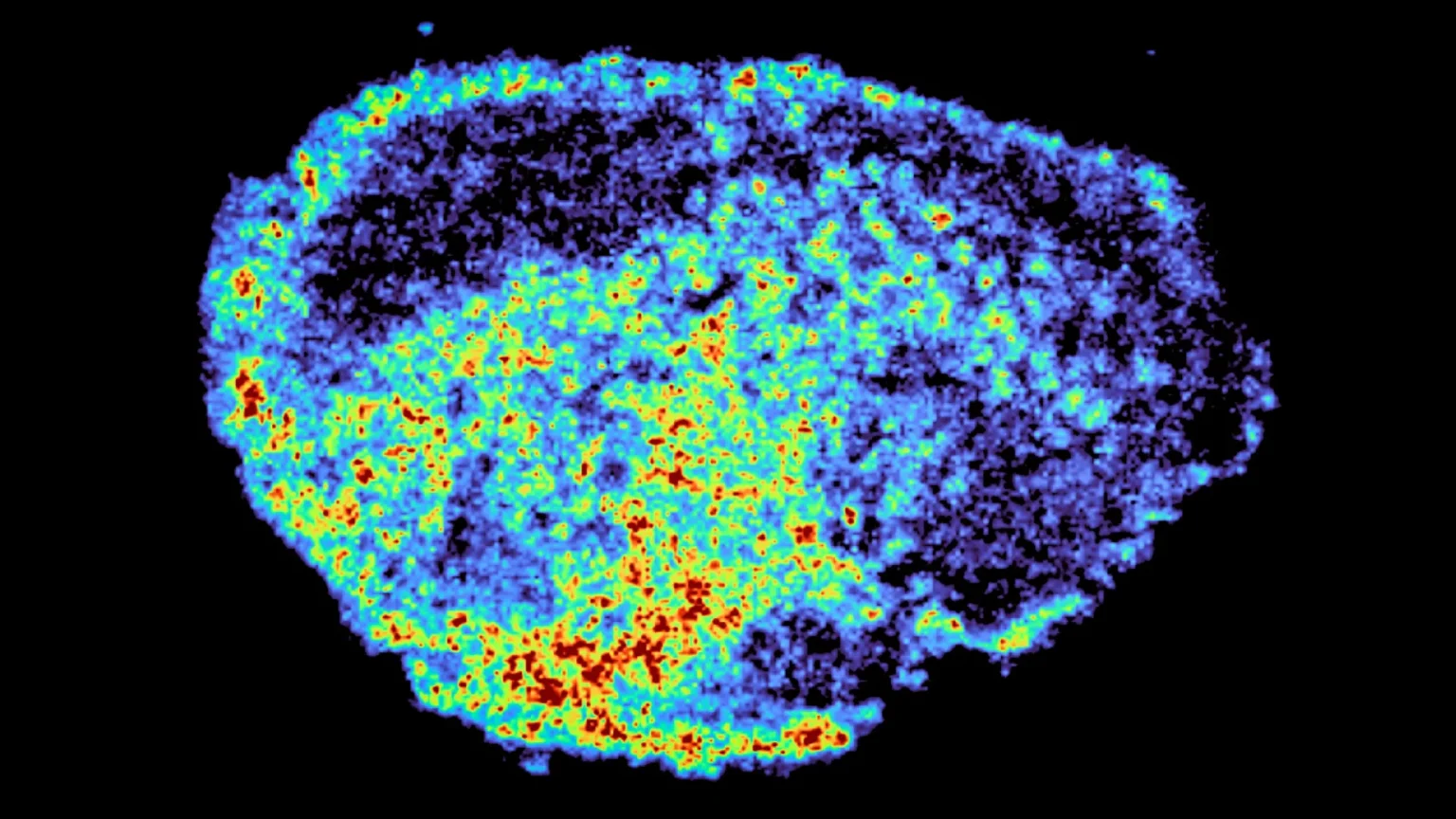
One of the biggest challenges in cancer treatment is that the same treatment can be very effective for some patients and not at all effective for others. New research published in nature communicationsA research team led by Dr. Louise Fetz from the MRC Institute of Medical Sciences (LMS) is taking a closer look at why this happens. The researchers focused on PARP inhibitors, a type of targeted cancer drug, and used advanced imaging tools to track how PARP inhibitors move through ovarian tumor samples.
Their findings indicate that these drugs can accumulate within lysosomes, small structures within cells that act as “recycling centers.” Once the drug is inside, it can become trapped and later released, affecting the performance of the treatment.
Mapping how cancer drugs spread within tumors
Cancer treatment options have expanded rapidly in recent years, improving outcomes for many patients. In particular, PARP inhibitors have revolutionized the treatment of ovarian cancer. However, not all patients benefit, and some develop resistance over time. For these drugs to work, they must accumulate within cancer cells at levels sufficient to cause cell death. Nevertheless, scientists still have a limited understanding of how drugs are distributed within tumors and what controls that process.
This study shows that efficacy depends not only on whether the drug reaches the tumor, but also on how it spreads within the tumor and within individual cells. To study this, the researchers used thin sections of ovarian tumors taken from patients and kept alive in the lab. These samples, known as “explants,” were treated with PARP inhibitors, allowing scientists to directly observe how the drug moves through real human tumor tissue.
The research team used mass spectrometry imaging to create a detailed map showing exactly where the drug accumulated. By combining this with spatial transcriptomics, they were able to examine gene activity in regions with high and low drug levels within the same sample. The results revealed significant differences in drug distribution both within individual tumors and between patients, even when using the same dose.
“A novel aspect of this study was that we used mass spectrometry imaging to directly measure and visualize drug uptake in patient tumor tissue. Through spatial mapping of drug molecules, we were able to use spatial transcriptomics to precisely identify areas of high and low drug concentration from the same tissue slice and compare gene expression,” said senior author and associate professor Dr Zoe Hall from Imperial’s Department of Metabolism, Digestion and Reproduction.
Lysosomes act as hidden drug storage
The researchers discovered that lysosomes play a central role in this uneven distribution. Some PARP inhibitors are drawn into and stored in these compartments, rather than being spread evenly throughout the cell. This creates an internal pocket where the drug accumulates.
These lysosomes act as sustained-release reservoirs, holding and gradually releasing drugs. This increases the amount of exposure for certain cells, while leaving other cells with much lower levels. Not all PARP inhibitors work the same. The study found that drugs such as rucaparib and niraparib are affected by this process, while others such as olaparib are not.
“We were surprised to find large variations in drug accumulation at the single-cell level. This variation was caused by the accumulation of drugs within lysosomes, which act as reservoirs and increase the exposure of cancer cells to drugs by storing and releasing drugs as needed,” says Carmen Ramirez Moncayo, Ph.D., first author and postdoctoral researcher at LMS.
What this means for the future of cancer treatment
PARP inhibitors are already widely used to treat ovarian, breast, and prostate cancer, and are being tested in many other types of cancer. A better understanding of how these drugs are stored and distributed within cells could lead to more personalized treatment strategies, improving efficacy while reducing resistance and relapse.
“By understanding how drugs are taken up by cells, we can understand whether this influences why anti-cancer drugs work for some people and not others. Ultimately, we hope to study the molecular signatures of patients’ tumors to be able to tailor treatment approaches in a more personalized way,” said senior author Dr. Louise Fetz, head of the Drug Transport and Tumor Metabolism Group at LMS.
This study was conducted using tumor tissue maintained outside the body. In real patients, since drugs are delivered via the bloodstream, tumor blood vessels are often disorganized, which may further contribute to the heterogeneity of drug distribution. Future studies will use animal models and larger patient groups to better understand how drug delivery, tumor architecture, and lysosomal storage interact in clinical settings, including in recurrent cancers.
This research was supported by funding from the Medical Research Council, Cancer Research UK, PhD students from the Integrative Toxicology Training Partnership managed by the MRC Toxicology Unit, and funding from the Victoria’s Secret Global Fund for Women’s Cancers Career Development Award in partnership with Pelotonia and AACR.