A newly designed microbial system turns waste plastic into a front-line Parkinson’s disease treatment, offering a glimpse into a future where waste becomes medicine.
Research: Microbial upcycling of plastic waste into levodopa. Image credit: jmcatholic / Shutterstock
In a recent study published in the journal natural sustainabilityresearchers have demonstrated that they have successfully engineered a biological process to “upcycle” poly(ethylene terephthalate) (PET) into levodopa (L-DOPA), a front-line treatment for Parkinson’s disease (PD). researchers fixed Escherichia coli Convert plastic-derived monomers into high-value pharmaceuticals under mild aqueous conditions.
This study overcomes critical biochemical hurdles involved in cellular trafficking and enzyme inhibition by separating the process across two cooperative microbial strains. This finding revealed that the optimized two-stage preparation system yielded a high production titer of 5.0 g L−1. Although this approach offers a potentially more sustainable route than traditional fossil fuel-derived chemical or chemoenzymatic synthesis, it remains a proof of concept rather than a fully optimized industrial process.
The plastic waste crisis and the challenges of sustainable chemistry
The modern chemical industry, particularly pharmaceuticals, is built on finite fossil resources, and despite saving millions of lives, this model is inherently unsustainable and has a negative impact on the environment. At the same time, the biosphere is under increasing stress from the global production and accumulation of plastics, mainly derived from fossil fuels.
According to environmental reports, more than 400 million tons of plastic are produced annually, of which around 360 million tons are disposed of as waste. Much of this waste is sent to landfills or incinerated, resulting in the loss of valuable carbon and the release of large amounts of greenhouse gases. While traditional recycling exists, researchers are increasingly turning to ‘upcycling’ – converting waste into higher-value products – as a more sustainable path towards a circular economy.
Levodopa (L-DOPA) is widely used as a treatment for Parkinson’s disease. Their commercial production typically relies on chemical or chemoenzymatic synthesis derived from fossil fuels, often involving harsh conditions and generating large amounts of waste.
Biological production of L-DOPA from glucose or amino acids has been investigated, but these approaches often have low efficiency and face challenges in terms of industrial scalability.
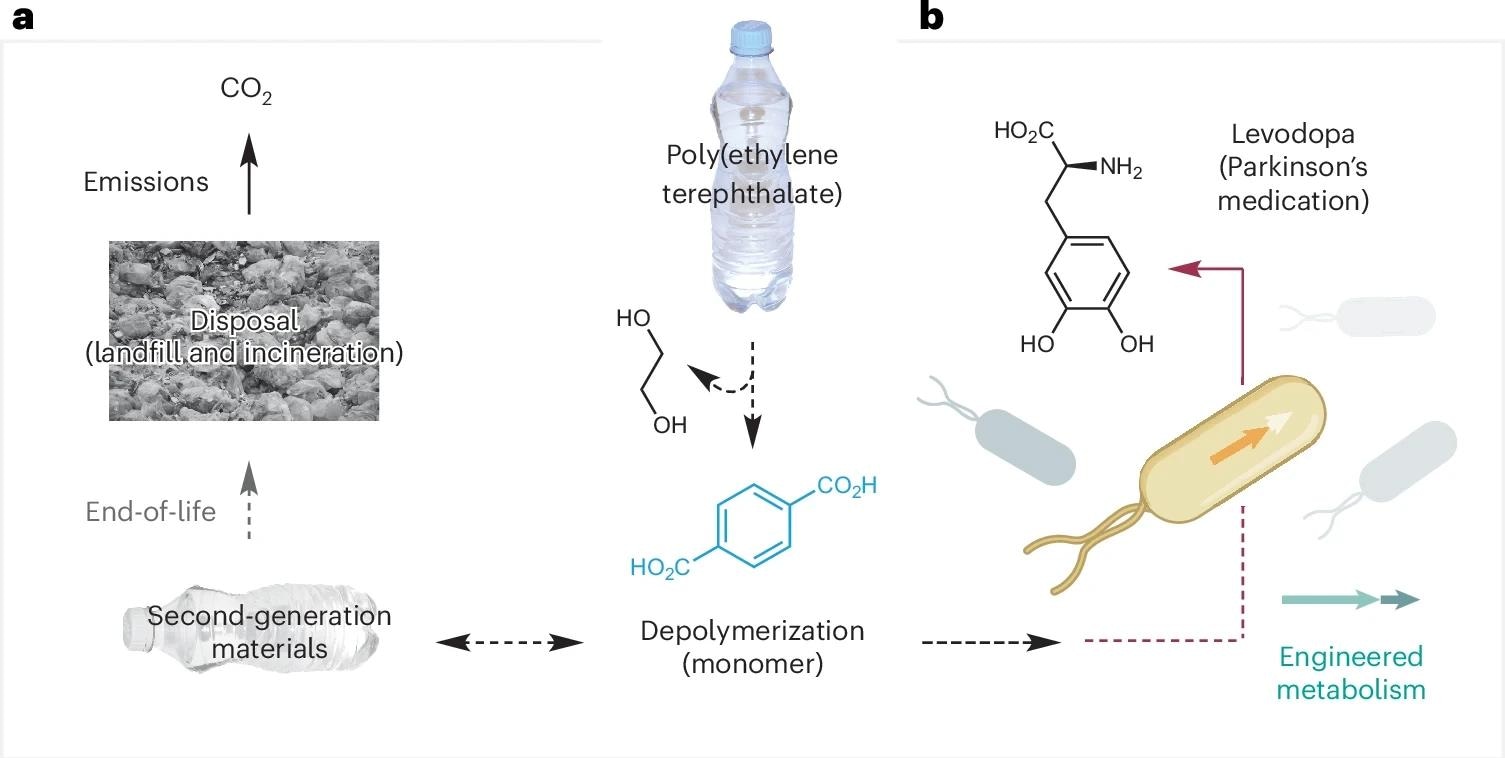
Approaches to recycling, upcycling, and environmental treatment of PET waste. This includes the proposed bioupcycling of PET waste into the Parkinson’s disease treatment l-DOPA using engineered bacteria. benow: Closed-loop recycling. bThis work: Upcycling of microorganisms. Credit: The photo is beRawpixel (https://www.rawpixel.com); Bacteria icon bBioicons (https://bioicons.com).
designed Escherichia coli Plastic conversion pathway
This study aimed to overcome these limitations by leveraging bioengineering strategies to transform plastic waste into complex therapeutic products. This approach focused on terephthalic acid (TPA), a monomer derived from the degradation of PET.
The researchers designed a new four-step biosynthetic pathway containing seven genes, and these genes were introduced. Escherichia coli BL21(DE3). Initial testing revealed two major bottlenecks.
First, bacteria had difficulty transporting TPA across the cell membrane at neutral pH. This problem was solved by expressing the transporter protein TpaK. Rhodococcus jostiiintake was significantly improved.
Second, the pathway intermediate protocatechuic acid (PCA) inhibited the last enzyme, tyrosine phenol-lyase (TPL), through feedback inhibition. In vitro experiments showed that PCA concentrations above 2 mM abolished detectable L-DOPA production, while conversion efficiency decreased from 80% to 0% above 1 mM PCA in whole-cell systems.
This challenge was overcome by splitting the pathway between the two microbial strains. One strain converts TPA to catechol and the second strain converts catechol to L-DOPA.
The system was also tested using real-world waste materials such as industrial hot stamping foil (HSF) and used plastic bottles. Furthermore, microalgae Chlamydomonas reinhardtii It was used to capture carbon dioxide (CO2) produced during the process, supporting a proof of concept for a carbon-neutral production cycle.
Experimental results and production efficiency indicators
The designed system achieved an L-DOPA titer of 5.0 g L−1 corresponding to a conversion efficiency of 84% from industrial waste in an optimized two-step workflow using foil-derived TPA. Addition of TpaK transporter significantly improved the conversion of TPA to PCA at neutral pH.
incorporate C. Reinhardti reduced culture headspace CO2 levels to undetectable levels within 12 h and demonstrated that metabolic byproducts were integrated into the biomass under experimental conditions.
Using TPA derived from discarded plastic bottles, the conversion rate was 49%. In another experiment with foil-derived TPA, this process yielded 193 mg of L-DOPA as a solid salt, equivalent to several clinical doses for early Parkinson’s disease.
Implications and limitations for sustainable pharmaceuticals
This study provides proof of concept that plastic waste can be converted into valuable pharmaceutical compounds and highlights a potential strategy to address both environmental pollution and sustainable pharmaceutical production.
However, further optimization is required before industrial application. Key areas include direct precipitation of L-DOPA from fermentation broths, removal of residual contaminants from plastic waste streams, genomic integration of pathway genes to eliminate the need for antibiotic selection, and further development of algal CO2 capture systems.