Cytotoxic T-lymphocytes act as specialized “killer” cells of the immune system, seeking out and eliminating infected and cancerous cells with remarkable precision. Its effectiveness relies on tightly controlled contact points called “immune synapses,” where it releases toxic molecules that destroy targets while keeping surrounding healthy cells intact. Until recently, scientists have struggled to observe the fine structure of this process in detail. Now, researchers from the University of Geneva (UNIGE) and Lausanne University Hospital (CHUV) have succeeded in visualizing these mechanisms in three dimensions under near-natural conditions. Their findings were; cell reportshed new light on how the internal organization of cytotoxic T cells supports their function and potentially advance research in immuno-oncology.
When the body faces an infection or cancer, cytotoxic T lymphocytes tightly bind to their targets and form an immune synapse. Through this special interface, it releases toxic substances that cause the destruction of harmful cells. This targeted approach allows the immune system to efficiently eliminate threats while minimizing collateral damage to nearby tissues.
Although scientists have long understood the basics of this process, studying its detailed structure at the nanometer scale inside intact human cells remains difficult. The big problem lies in how the samples are prepared, as traditional methods can distort delicate cellular components. Existing imaging techniques often require researchers to choose between high resolution, wide field of view, or preservation of the natural structure of cells.
Cryo-expansion microscopy reveals hidden details
To overcome these challenges, the UNIGE and CHUV-UNIL teams, with support from the ISREC Foundation TANDEM program, used an advanced technique called cryo-expansion microscopy (cryo-ExM). “This technique freezes cells very quickly and instantly, placing them in a so-called glassy state, where the water solidifies without forming crystals, thus preserving the biological structure faithfully. The sample is then physically expanded using absorbable hydrogels, making it possible to observe the internal organization with great precision while preserving its near-natural structure,” explains Virginie Hamel, Senior Lecturer in the Department of Molecular and Cell Biology at UNIGE’s Department of Physics.
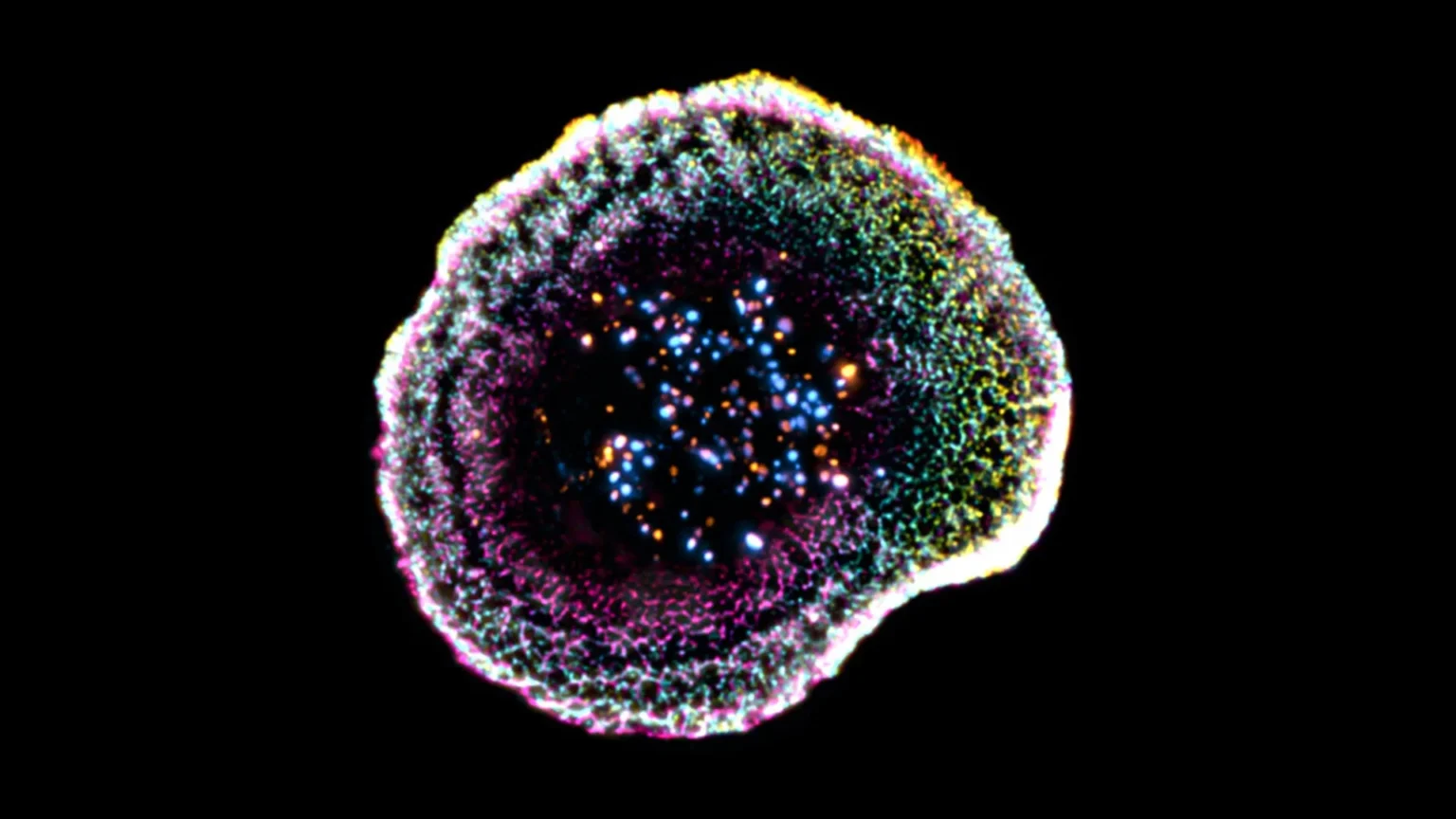
Using this approach, the researchers discovered new structural features at the point where immune cells meet their targets. “Our study revealed that at the point of contact between an immune cell and its target, the membrane forms a kind of dome. That structure seems to be related to adhesive interactions and the internal organization of the cell,” said Florent Lemaitre, a postdoctoral researcher in the Department of Molecular and Cell Biology at UNIGE’s Department of Science and lead author of the study. The research team also examined with unprecedented clarity the cytotoxic granules responsible for killing target cells. They found that these granules can vary in structure and in some cases contain one or more “cores” in which the active molecules are concentrated.
From experimental cells to actual tumors
The researchers extended their method beyond isolated cells and applied it directly to human tumor samples. “We have extended this approach to human tumor tissue, allowing us to directly observe tumor-infiltrating T lymphocytes and their cytotoxic mechanisms at the nanometer scale. This allows us to directly study the immune response in a clinical setting and better understand the mechanisms that determine its effectiveness,” explains Benita Wolf, chief resident and associate investigator in the CHUV Department of Clinical Oncology, who co-led the study.
This study provides a valuable framework for studying immune responses in real-world situations by providing a three-dimensional, near-native view of how these immune cells function. These insights may help improve our understanding of what drives and limits successful immune attacks against cancer, and improve treatments, particularly in tumor immunology.