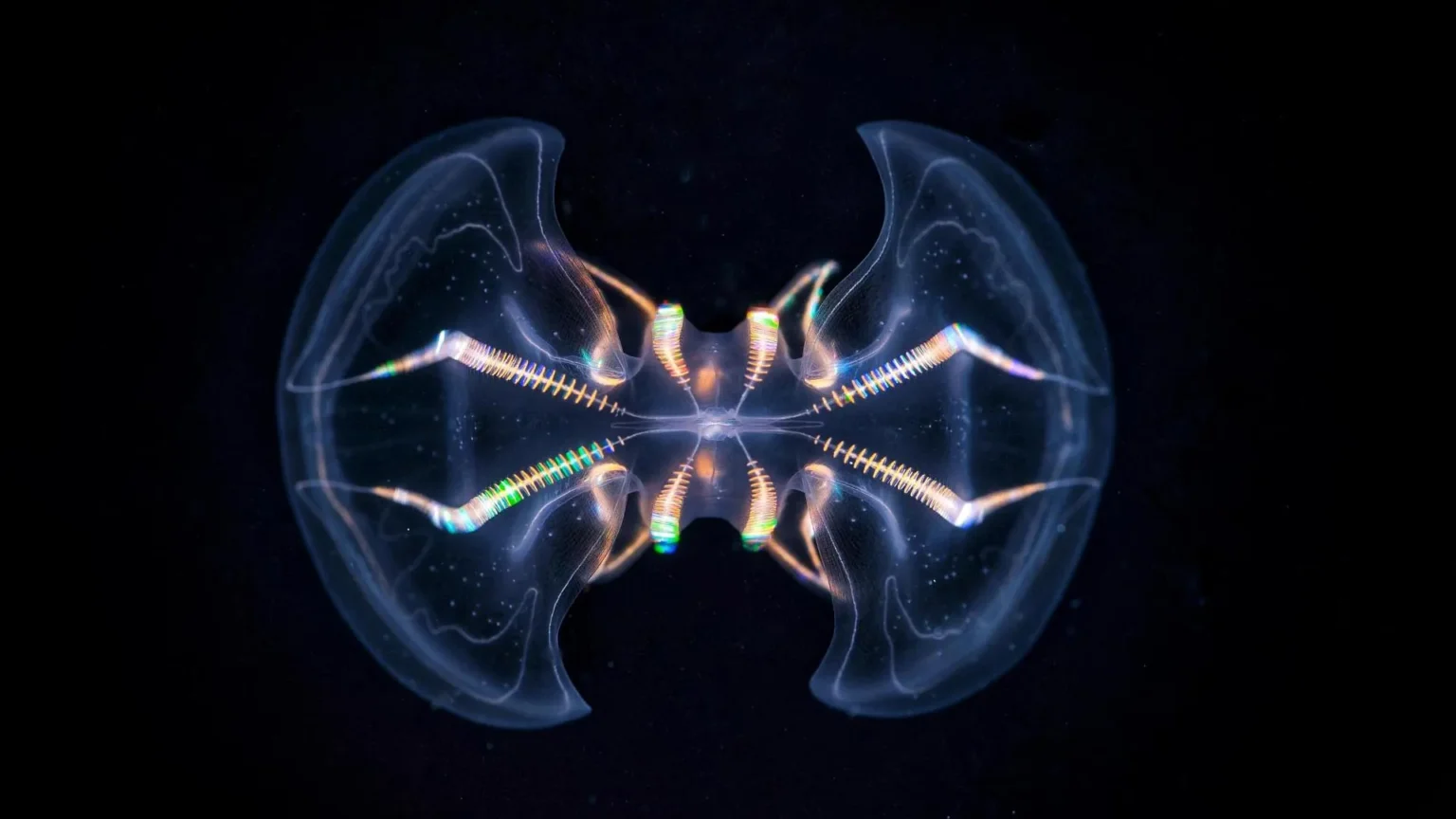
Detailed three-dimensional reconstruction of the major sensory structures of ctenophores reveals much greater structural and functional complexity than scientists previously realized. The results suggest that some of the earliest animals may have had simple brain-like systems, providing new insights into how nervous systems evolved.
Ctenophores (commonly known as comb jellies) are delicate gelatinous marine animals that appeared in Earth’s oceans about 550 million years ago. These organisms have specialized sensory structures called abdominal organs (AOs) that allow them to detect gravity, pressure, and light. New morphological research presented in scientific progress This indicates that this organ is significantly more complex than previous studies suggested.
“We have shown that the AO is a complex and functionally unique sensory system,” said Pawel Burkhardt, group leader at the Michael Saas Center at the University of Bergen. “Our study significantly increases our understanding of the evolution of behavioral coordination in animals.”
Mapping the cellular structure of ancient organs
To understand how abdominal organs are organized internally, the researchers, in collaboration with Maike Kittelmann from Oxford Brookes University, used advanced volumetric electron microscopy. This approach allowed us to create highly detailed three-dimensional reconstructions of the structure.
The analysis revealed 17 different cell types within the ventral organ, including 11 secretory and ciliated cell types that have never been identified before. This wide variety of cells confirms that the AO functions as a sophisticated multimodal sensory organ.
“I was immediately struck by the morphological diversity of abdominal organ cells. Working with volumetric EM data feels like discovering something new and exciting every day,” said Anna Ferraioli, a postdoctoral fellow at the Michael Szasz Center and lead author of the study. “The AO is surprisingly complex when compared to the apical organs of cnidarians and bilaterians. It’s so unique!”
Hybrid neural communication system
Beyond cellular diversity, the ventral organ also appears to be closely related to the comb jelly’s nervous system. Ctenophores have neural networks composed of fused neurons that form continuous structures throughout their bodies.
The researchers discovered that this neuropil forms direct synaptic connections with cells in abdominal organs, creating a pathway for two-way communication. At the same time, many cells within the AO contain large numbers of vesicles, indicating that they may release a wide range of chemical signals through a process known as volume transfer. Together, these mechanisms suggest that this organ relies on both synaptic and nonsynaptic signaling.
“I think our study provides an important perspective on how much we can learn from studying morphology,” Ferraioli explains. “The AO is definitely not similar to our brains, but it could be defined as an organ that ctenophores use as a brain.”
Clues about brain evolution
The research team also looked at how certain developmental genes are expressed in ctenophores. Many genes involved in the formation of body tissues in other animals are present in these organisms, but their expression patterns differ significantly.
This difference suggests that the ventral organ may not be directly equivalent to the brain found in other animal groups. “In other words, it appears that the central nervous system was invented multiple times during evolution,” Burkhardt added.
Linking neural structure and behavior
Further support for these findings comes from related research led by Kei Shirokura of Japan’s National Institute for Basic Biology and Professor Gaspard Gékely of the University of Heidelberg. In a separate study in which Burkhardt also participated, scientists reconstructed the complete neural wiring of the comb jellies’ gravity-sensing organs.
By combining high-speed imaging and three-dimensional reconstruction of more than 1,000 cells, the researchers showed how a network of fused neurons coordinates the beating of cilia in different parts of the animal’s body. This adjustment allows the comb jellyfish to maintain its orientation as it moves through the water.
“Similarities to neural circuits in other marine organisms suggest that a solution comparable to gravity sensing may have evolved independently in distant animal lineages,” Jokura said.
Reconsidering the origin of the nervous system
Together, these studies suggest that early nervous systems may have been more concentrated than scientists previously thought. Ferraioli said the next stage of the research will focus on identifying the molecular signatures of the newly discovered cell type and investigating how strongly ventral organs influence comb jelly behavior.