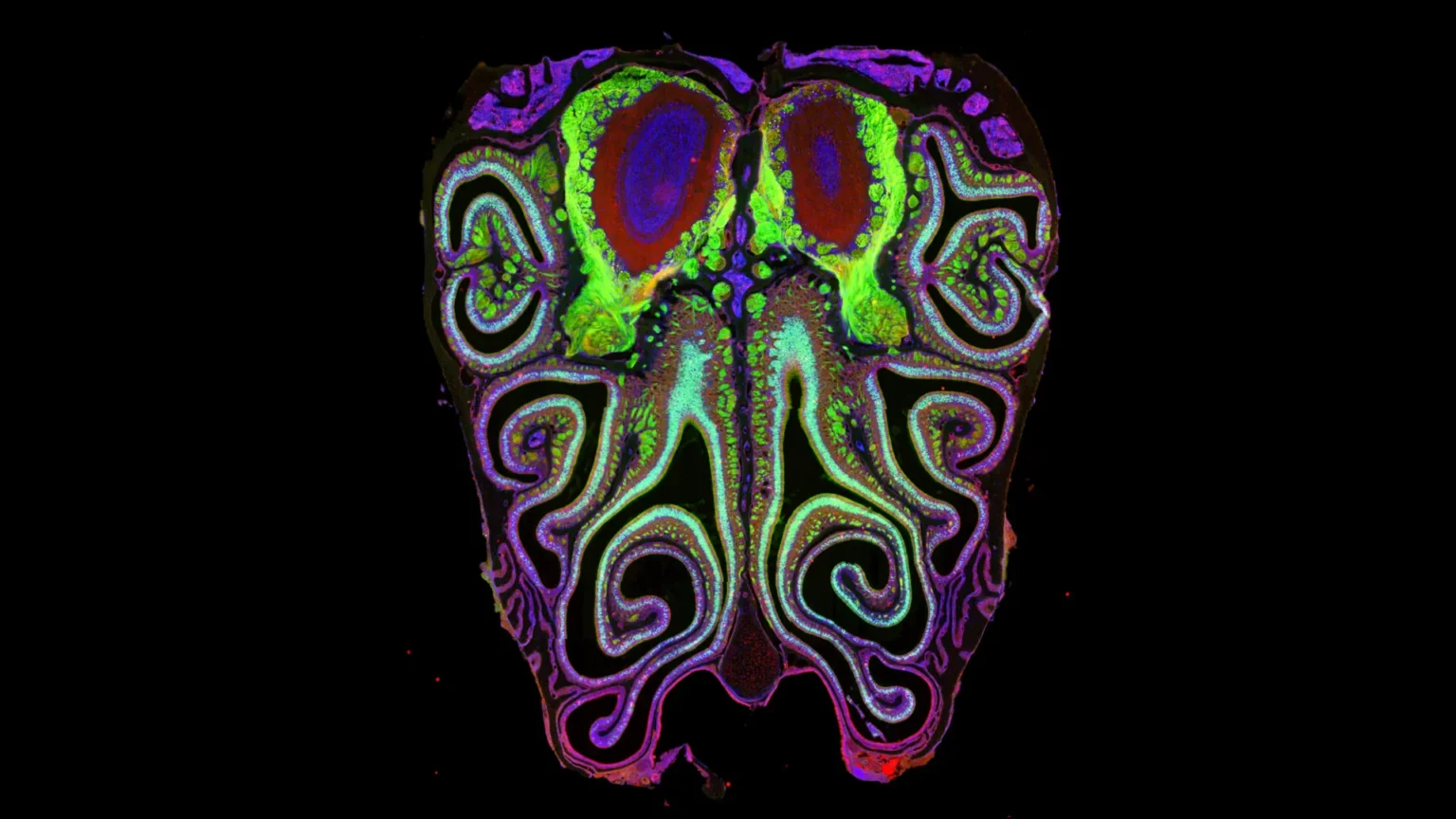
Scent shapes how we experience the world every day. It helps detect danger, adds depth to flavor, and is strongly associated with memories and emotions. Despite its importance, scientists have struggled to fully understand how this sense works at a biological level.
“The sense of smell is very mysterious,” says Sandeep (Robert) Dutta, a professor of neurobiology at Harvard Medical School’s Blavatnik Institute. Compared to vision, hearing, and touch, the basic biology of smell remains poorly understood.
Scientists create first detailed map of odor receptors
In a new study in mice, Dutta and his colleagues have created the first detailed map showing how more than 1,000 different scent receptors are arranged in the nose.
What they discovered overturned long-held assumptions. The neurons carrying these receptors are not randomly distributed but are highly organized. They are grouped by receptor type and form horizontal bands, or stripes, that run from the top of the nose to the bottom.
“Our results bring order to a system previously thought to lack order and conceptually change the way we think this works,” said Datta, the study’s senior author.
The researchers also showed that this map of the nose matched the corresponding map of the brain’s olfactory bulb. This connection provides new insights into how scent information travels from the nose to neural circuits.
The findings were published on April 28th. cell.
Long search for odor maps
Scientists have long understood how sensory receptors are arranged in the eyes, ears, and skin, and how those patterns are connected to the brain. Smell is an exception.
“The sense of smell is the only exception. It’s a sense that hasn’t been on the map for a long time,” Datta said.
One reason is complexity. Mice have approximately 20 million olfactory neurons, each of which expresses one of more than 1,000 types of receptors. In contrast, human color vision relies on only three major receptor types. Each odor receptor detects a specific set of odor molecules, making the system more complex.
Researchers began identifying odor receptors in 1991. Over the next few decades, researchers explored patterns in how these receptors are arranged. Previous studies have suggested that the receptors occur only in a small number of widespread zones, leading to the idea that their placement is largely random.
As new genetic tools became available, Datta’s team revisited the problem in a more powerful way.
Hidden patterns revealed in millions of neurons
The research team analyzed approximately 5.5 million neurons from more than 300 mice. They combined single-cell sequencing, which identifies the receptors each neuron expresses, with spatial transcriptomics, which pinpoints where those neurons are located.
“This is probably the most sequenced neural tissue ever, but we needed data of that scale to understand the system,” Datta said.
Their results revealed a clear and consistent pattern. Neurons form tightly organized overlapping horizontal stripes based on the receptors they carry. This arrangement was nearly identical in all animals studied, and roughly corresponded to how odor information is mapped in the brain.
How to form odor maps
The researchers also investigated how this precise structure develops. They identified retinoic acid, a molecule that regulates gene activity, as a key factor.
A gradient of retinoic acid in the nose appears to guide neurons, helping each neuron activate the correct odor receptors depending on its location. When the researchers changed the levels of this molecule, the entire receptor map shifted up or down.
“We have shown that development can accomplish this feat of organizing a thousand different olfactory receptors into an incredibly precise map that is consistent across animals,” Datta said.
Another study published in the same issue of the journal Cell and led by the lab of Catherine Dulac, a Zander professor in Harvard’s Department of Molecular and Cellular Biology, found consistent findings.
What this means for treating anosmia
This discovery could have practical implications beyond basic science advances. There are currently few effective treatments for anosmia, despite its potential to impact safety, nutrition, and mental health.
“You can’t modify smell unless you understand how it works at a fundamental level,” Dutta says.
The research team is now working to understand why receptor stripes appear in the particular order and whether the same organization exists in humans. This knowledge could lead to new approaches aimed at restoring the sense of smell, such as stem cell therapy and brain-computer interfaces.
“Smell has such a deep and far-reaching impact on human health that restoring it is important not only for pleasure and safety, but also for psychological well-being,” Datta said. “If we don’t understand this map, we are doomed to fail in developing new treatments.”
Authors, funding, and disclosures
Additional authors on this paper include David Brann, Taya Tsukahara, Cyrus Tau, Dennis Kalloor, Rylin Lubash, Lakshanya Kannan, Nell Klimpert, Mihaly Kollo, Martin Escamilla-Del-Arenal, Bogdan Bintu, Andreas Schaefer, Alexander Fleischmann, and Thomas Bozza.
Funding for the research was provided by the National Institutes of Health (grants R01DC021669, R01DC021422, R01DC021965, and F31DC019017), the Yang Tang Collective at Harvard University, and a National Science Foundation Graduate Research Fellowship.