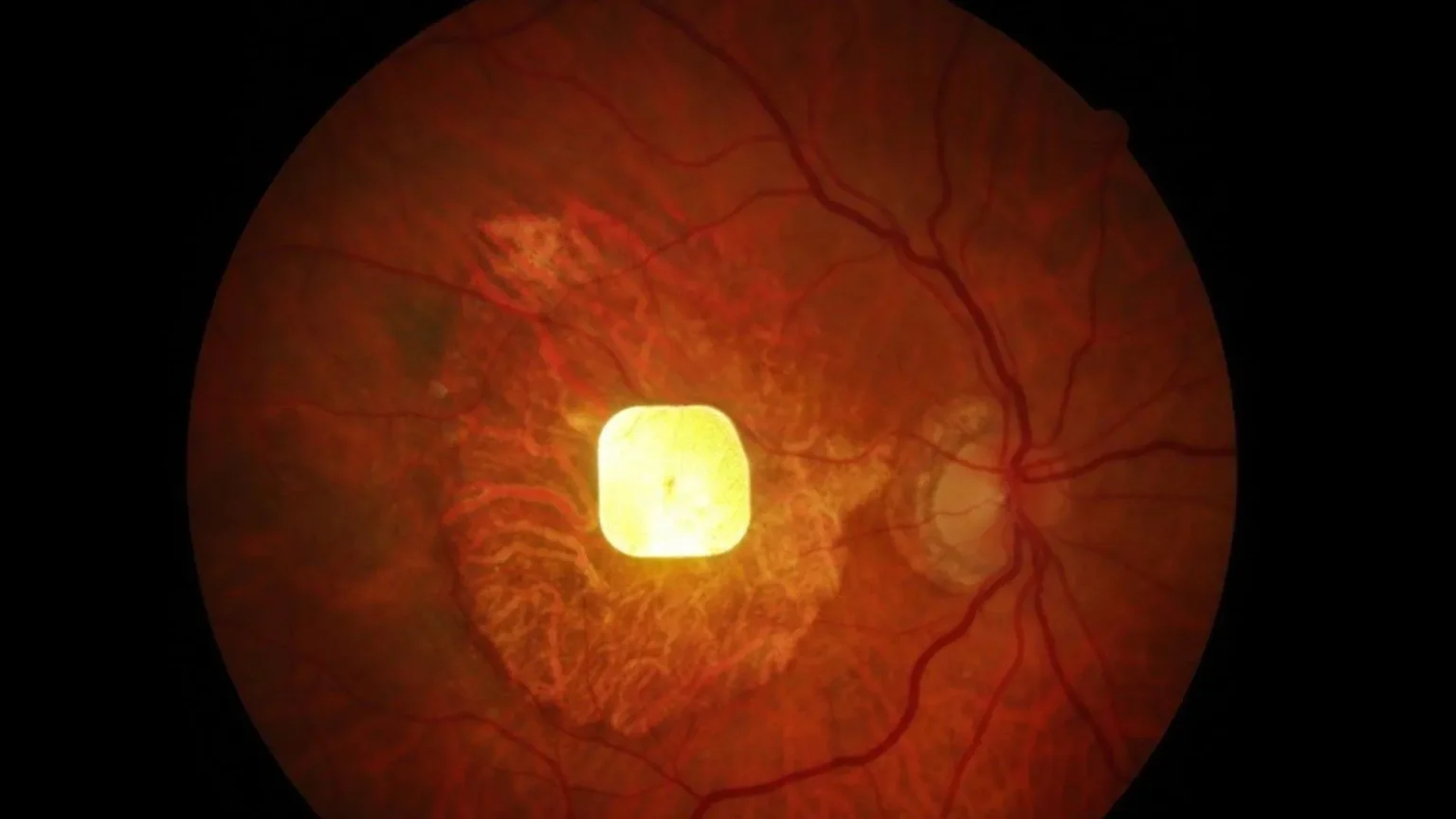
A small wireless retinal implant helps restore central vision in patients with progressive age-related macular degeneration (AMD), according to results published today. New England Medical Journal. Progressive dry AMD, also known as geographic atrophy (GA), is the most common cause of permanent blindness in older adults, affecting more than 5 million people worldwide.
This international, multicenter clinical trial was co-led by Jose Alain Sahel, MD, director of the UPMC Vision Institute. Dr. Daniel Palankar, Professor of Ophthalmology, Stanford University; Dr. Frank Holz, professor of ophthalmology at the University of Bonn in Germany.
Clinical trials showed significant improvement in visual acuity
Of the 32 participants who completed the 1-year follow-up, 26 (81%) experienced significant improvement in visual acuity. Additionally, 27 participants (84%) said they read numbers and words at home using the artificial vision provided by the device.
With the implant, patients were able to score an average of 25 letters (equivalent to about 5 lines) on a standard eye chart. Overall, 81% of participants improved by at least 10 letters.
“This is the first time that a vision restoration attempt has achieved these results in a large number of patients,” said Sahel, lead author of the study and chair of the Department of Ophthalmology at the University of Pittsburgh School of Medicine. “More than 80% of our patients can read and some even read the pages of a book. This is something we could never have dreamed of when we started this journey with Daniel Palankar 15 years ago.”
How PRIMA retinal implants work
As AMD progresses, the light-sensing cells in the retina are permanently damaged, causing central vision to become blurry. In a healthy eye, these photoreceptors capture light and convert it into electrical signals. These signals travel through nerve cells in the back of the eye and through the optic nerve to the brain, where an image is formed.
Palancar’s proprietary PRIMA system is designed to replace damaged photoreceptors with a 2×2 mm wireless implant. The device converts light into electrical signals that stimulate the surviving retinal cells. A camera built into special glasses records images and transmits them to the implant using invisible near-infrared light. The implant then converts that light into electrical pulses, restoring the transmission of visual information to the brain. Users can adjust zoom and contrast settings to improve visibility.
International learning across Europe
The PRIMAvera trial enrolled 38 participants aged 60 years and older at 17 medical centers in five European countries: France, Germany, Italy, the Netherlands, and the United Kingdom.
After 12 months of use, all side effects associated with this procedure have disappeared. Most participants showed a clear improvement in their ability to identify letters on an eye chart. One person improved by 59 characters, which is equivalent to 12 lines.
“Although it is not yet possible to fully restore 20/20 vision with implants alone, at UPMC we are researching ways to further improve people’s quality of life and move them beyond the threshold of legal blindness,” Sahel said.
Regulatory actions and ongoing research
Following these results, device manufacturer Science has submitted an application seeking approval for clinical use in Europe and the United States. UPMC became the first U.S. center to implant the PRIMA device in 2020, thanks to a study led by Joseph Martell, MD, associate professor of ophthalmology.
Additional researchers participating in this study represent the University of Bonn. Adolphe de Rothschild Foundation Hospital and the 15-20 National Eye Hospital in Paris. Moorfields Eye Hospital, London. University of Rome Tor Vergata, etc.
This research was funded by Science Corporation, Alameda, California (formerly Pixium Vision SA, Paris, France).