Every second, countless electrical charges pass through the human body. These subtle signals are essential to life. They facilitate communication between cells, enable energy production, and support metabolism. None of these work without the carefully controlled movement of charge across the cell membrane and within the cell. In many ways, charge transport serves as a fundamental control system in biology.
Phosphoric acid (H3PO4) and related compounds are present almost ubiquitously in biological systems. They are important components of DNA and RNA, part of cell membranes, and the center of ATP, the molecule that stores and transfers energy within cells. These compounds play a major role in the transfer of positive charges, also known as protons. Because of phosphoric acid’s excellent ability to conduct protons, it is widely used beyond biology in technologies such as batteries and fuel cells.
How protons pass through molecules
Protons move through phosphate-containing materials in a unique way. They do not move freely, but instead jump from one molecule to another. Hydrogen bonds act as pathways to guide this movement. This process, known as “proton shuttling,” allows charges to move very quickly.
Scientists have long understood that proton shuttling occurs, but the exact molecular details remained unclear. To investigate this, researchers from the Fritz Haber Institute’s Department of Molecular Physics, together with collaborators in Leipzig and the United States, focused on the key molecular structures involved in this process. Their goal was to determine how the early stages of proton transfer actually occur.
Research on proton transport in extreme cold
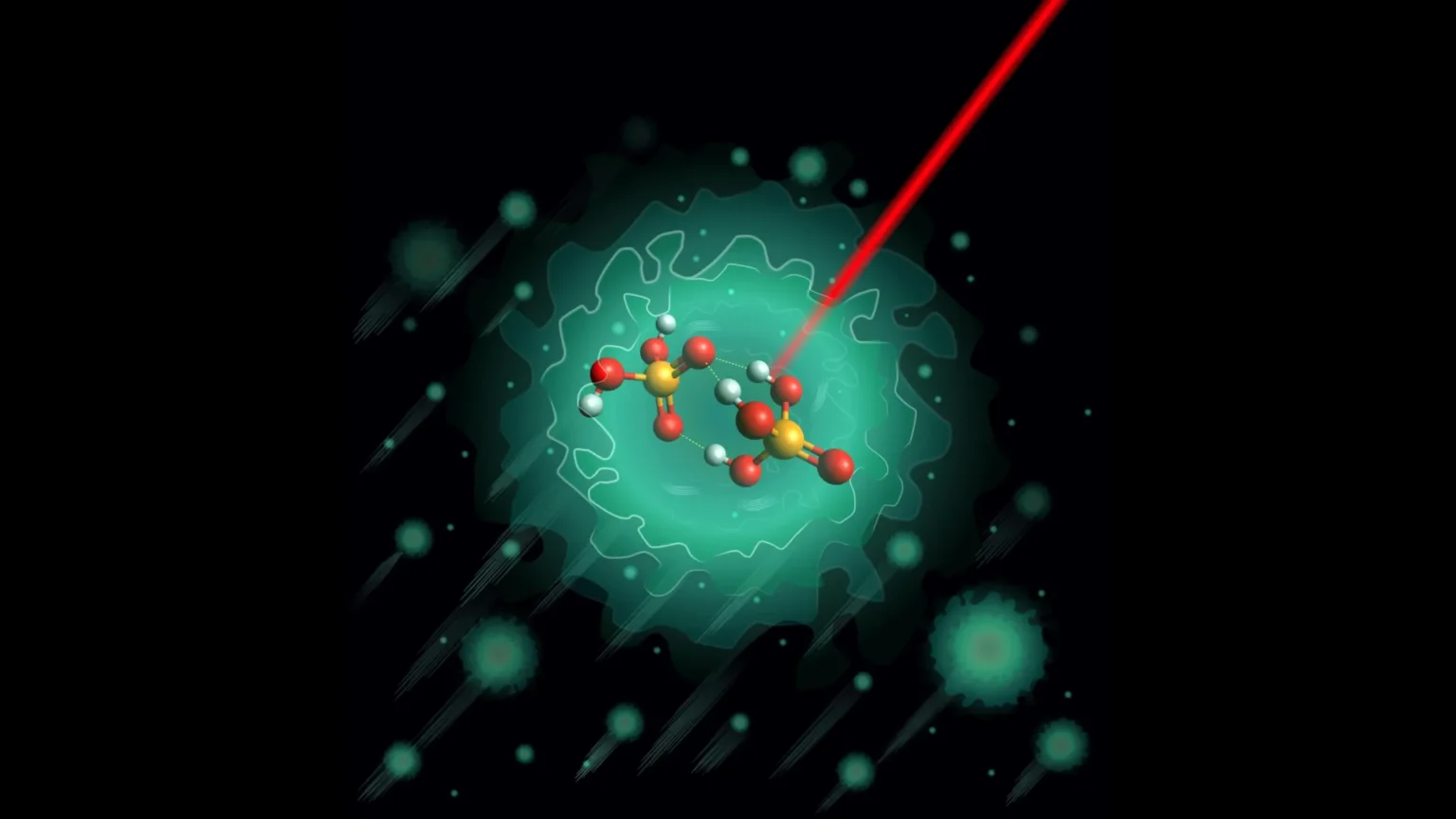
Previous research suggested that certain negatively charged molecules can initiate the proton shuttling process. This molecule, known as the deprotonated dimer H3PO4.H2PO4-, was the focus of the study.
To find out more, scientists created the molecules in the lab and cooled them to extremely low temperatures. By placing it inside helium nanodroplets, they lowered its temperature to just 0.37 degrees above absolute zero. At this temperature, unnecessary disturbances are almost completely eliminated. This allowed the researchers to analyze its structure with high precision using infrared spectroscopy.
The experimental results were combined with quantum chemical calculations that helped predict how the molecules would arrange and behave. Combining these approaches provides a clearer picture of molecules than using either method alone.
A single structure appears
The results revealed an unexpected result. Theoretical models predicted that the molecule could exist in two similar structures. However, the experimental data showed only one stable configuration.
This structure is relatively rigid and features three hydrogen bonds connected through a shared oxygen atom. There are also high barriers that limit the ease with which protons can move. Similar binding patterns have been observed in other phosphate clusters, suggesting that this arrangement may be a common structural feature.
These results highlight an important point. Even sophisticated theoretical models can miss important details, so experimental validation is essential to understanding molecular structure.
Why is this discovery important?
This study helps explain the molecular basis of phosphoric acid’s remarkable proton conductivity, often described as “nature’s proton highway.” By identifying a single, well-defined structure of the major anionic dimer H3PO4·H2PO4-, scientists are now able to better understand how protons move through these systems.
This discovery also provides a valuable reference point for improving quantum chemical models of phosphate-based molecules. Additionally, it could lead to the development of new materials with enhanced proton conductivity, which is important for technologies such as fuel cells. At the same time, this study deepens our understanding of how proton transfer works in biological systems.
Main highlights
- What they learned: The researchers focused on a pair of phosphate molecules known as ionic dimers. This small but powerful system plays a critical role in transporting positive charges within living organisms and is also widely used in technologies such as fuel cells. The goal was to understand what makes it so incredibly efficient at transporting charge.
- How they studied it: To capture extremely clear images of the molecules, the researchers cooled them to an extreme temperature of just 0.37 Kelvin. At this near-absolute-zero temperature, the researchers used a combination of infrared spectroscopy and quantum chemical calculations to map its structure with great precision.
- What they discovered: Instead of finding two possible structures as predicted by theory, experiments revealed only one stable form. This structure features a specific hydrogen bond arrangement that closely matches patterns seen in other phosphate systems, suggesting a common structural design.
- Why it’s important: This discovery helps explain the molecular basis of nature’s proton highway, a process that allows phosphates to conduct protons very efficiently. This deeper understanding could lead to the development of better energetic materials and improve our knowledge of how charge transfer works within living systems.