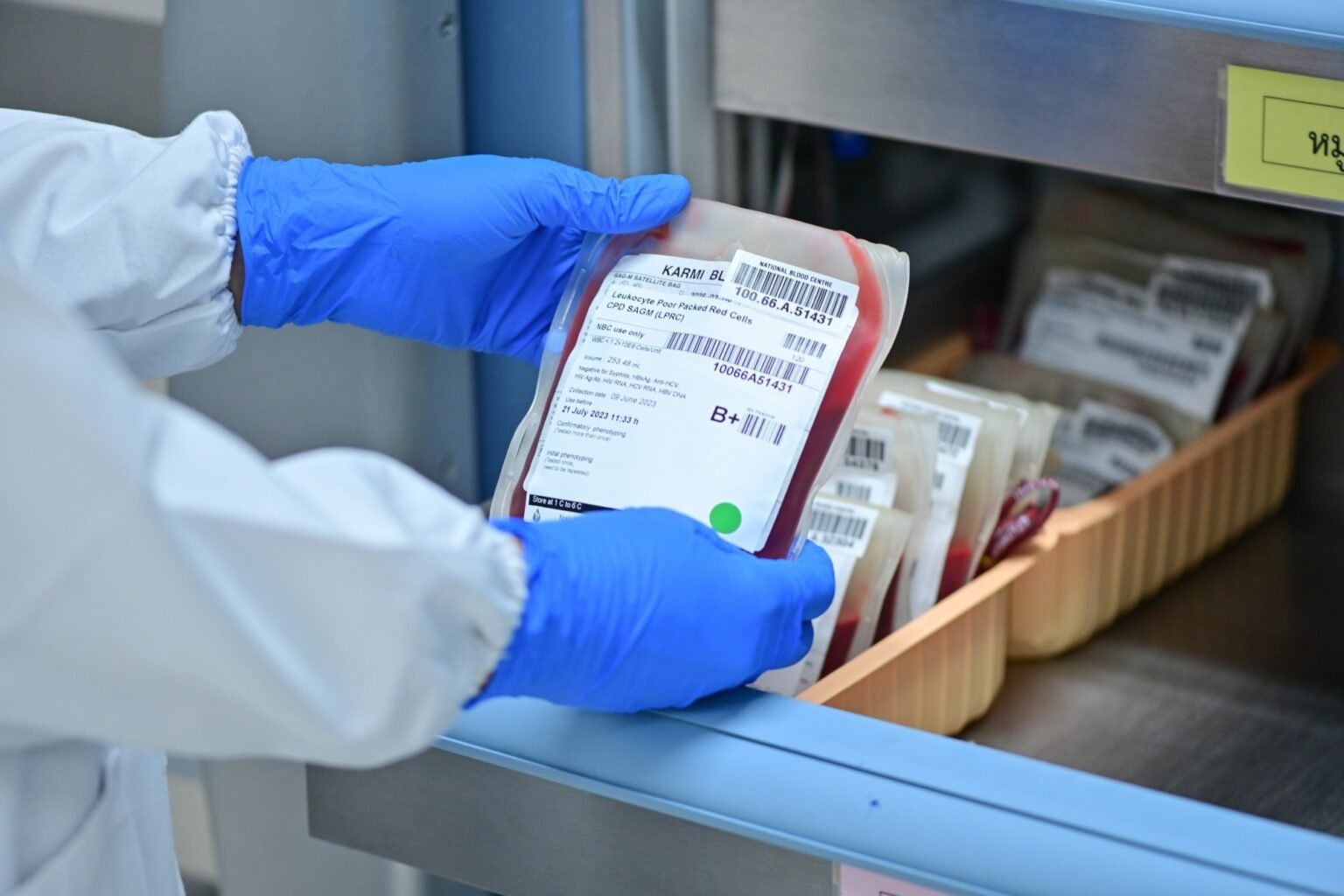
The increasing demand for “unvaccinated” blood poses challenges to modern blood transfusion systems. Real-world data now reveals how these demands can delay care, strain resources, and put vulnerable patients at risk.
 Study: Directed donation of unvaccinated blood: deviations from evidence-based medicine associated with clinical harm, wasted resources, and surveillance gaps in a 2-year single-center series. Image credit: Anakis srk/Shutterstock.com
Study: Directed donation of unvaccinated blood: deviations from evidence-based medicine associated with clinical harm, wasted resources, and surveillance gaps in a 2-year single-center series. Image credit: Anakis srk/Shutterstock.com
Requests for “unvaccinated” blood are on the rise, despite clear opposition from regulators and professional bodies. research in transfusion These demands are associated with delays in treatment, worsening of clinical symptoms in some patients, and inefficient use of health care resources.
Direct donations resulting from safety concerns
Blood transfusions are a fundamental part of modern medicine. Its safe and effective use relies on an anonymous, voluntary blood donation system based on altruism and community spirit. This model is supported by rigorous donor screening, infectious disease testing, and rigorous quality control measures.
Another approach is direct blood donation, where a specific donor donates blood to a designated recipient. This is usually reserved for rare situations, such as when a patient has an unusual blood type or there are no compatible donors in the area. But direct donations have also been used in other settings, such as during the early days of the human immunodeficiency virus (HIV) epidemic, when fears of infection from unknown donors led to a surge in such requests.
In response, screening protocols have been strengthened and nucleic acid tests have been introduced to improve the detection of viral infections. The risk of transfusion-associated transmission of major viruses is currently estimated to be less than 1 in 1 million.
Recent trends target ‘vaccinated’ blood
The coronavirus disease 2019 (COVID-19) pandemic has made the practice popular again due to the misconception that blood from vaccinated donors may be unsafe.
Regulators and professional bodies have strongly opposed such requests, saying blood centers do not track COVID-19 vaccination status. Additionally, available evidence supports the safety of blood from vaccinated donors.
Nevertheless, some patients remain convinced and continue to request direct donations from unvaccinated donors.
Real case series
The current study considers the real-world implications of responding to such requests outside of standardized consultation channels, building on prior literature that primarily addresses this issue from a theoretical or ethical perspective, although data on clinical outcomes are limited.
The authors report on a 2-year period in which standard blood transfusions at the Vanderbilt University Medical Center (VUMC) blood bank were refused because the blood was recognized as coming from a vaccinated person. They evaluated downstream effects on clinical outcomes, blood utilization, and healthcare operations.
During this period, blood banks processed 144,856 units, of which only 0.03% (48 units) were directly donated. These were all requested due to concerns about vaccinated donors and involved 15 patients, nine of whom were children.
These cases spanned a variety of clinical scenarios, including elective major surgery, critical illness, transplantation, and cancer treatment. Involvement of ethics and transfusion medicine experts was rarely documented, highlighting significant gaps in oversight.
Directed Donation Fate
Of these patients, 13 received one or more direct donor units. Two patients did not receive transfusions after the red blood cells arrived, despite being provided with five red blood cells, and no adverse outcomes were reported.
Overall, 31 of the 48 units were transfused to the intended recipient. Of the remaining units, most were placed in standard inventory, but three were placed in cold storage for intended recipients and two expired and were discarded before use.
Adverse clinical effects
In four cases, refusal of standard blood products by surrogate mothers (usually parents or guardians who make medical decisions on behalf of patients who are too young or unable to consent) was associated with adverse outcomes. Two children requiring cardiovascular surgery experienced delays or cancellations, and one patient developed hemodynamic shock as his hemoglobin level fell to 3.6 g/dL.
In another case, a delay in transfusion caused hemoglobin to drop to 5.9 g/dL, which was then transfused at 9.2 g/dL, above institutional guidelines, to avoid wasting the indicated units.
Immediate safety concerns
Transfusion medicine experts play a critical role in guiding safe blood transfusion practices, assessing the risk of infection, and recommending evidence-based alternatives to directed blood donation. However, in this series, many requests bypassed these standard safety routes. In contrast, other facilities require formal involvement of transfusion medicine experts before proceeding with such requests.
In most cases, surrogate decision-making was used, with parents acting on behalf of children who were unable to consent.
“Blood supply safety is premised on multi-layered risk mitigation, and no single layer, including testing, is assumed to be foolproof.” Directed donations circumvent basic safeguards of anonymous, voluntary donations and may therefore have a less favorable donor profile than community donations.
All transfused patients in this series received at least one unit of blood from a family member. First-time donors, who are more common in direct blood donation, have a slightly higher residual risk of transfusion-related infection, although the absolute risk remains very low.
In some cases, parents may unintentionally underreport behaviors that increase the risk of infection based on the mistaken belief that their blood is inherently safe for their child.
Direct family donations also carry the risk of transfusion-associated graft-versus-host disease (TA-GVHD). TA-GVHD is a rare but potentially fatal complication that requires irradiation of blood products before transfusion. This adds complexity, cost, and additional processing steps that may be overlooked if the request proceeds without expert input.
long term problems
Directed donations can also have long-term effects. Recipients may develop immune sensitization to family members who may eliminate the individual as a future organ or stem cell donor.
Another concern is transfusion-associated acute lung injury (TRALI). This is a serious and potentially fatal reaction associated with maternal blood transfusion in children. This occurs when the mother’s antibodies formed against the father’s antigens during pregnancy trigger an immune response in the recipient. Modern blood transfusion practices are designed to minimize this risk.
Impact on third parties
Unused indication units are often released into the general blood supply. However, neither clinicians nor the ultimate recipients are usually aware that these units come from a donor group with a more unfavorable risk profile than standard repeat volunteer donors. This effectively transfers potential risk from consenting family members to unrelated patients and raises ethical concerns regarding the principle of doing no harm.
Future direction
A series of cases suggests that these requests are driven by concerns about the use of vaccinated blood and are associated with delays in treatment, possibly worsening of clinical symptoms, and inefficient use of resources. In some cases, patients’ conditions deteriorated while waiting for the unit to arrive.
To avoid this situation, the authors recommend establishing a structured consultation workflow for all directed donation requests and replacing the optional online submission pathway with a mandatory transfusion drug consultation before proceeding with blood collection. These should include bloodless medicine specialists and transfusion medicine specialists.
Facility policies should be revised to protect clinicians and patients alike and prevent directed donations for reasons other than safety. Additionally, we recommend developing different pathways depending on the nature of your needs.
Finally, public education is essential to correct misconceptions about the safety of blood transfusions based on COVID-19 vaccination history.
Health systems should implement standardized counseling, documentation, and escalation pathways that are consistent with existing guidance.
The authors note that the study is limited by its small size, by only including cases in which the directed force reached the blood bank, and by its observational design, which means a causal relationship cannot be clearly established.
Click here to download your PDF copy.
Reference magazines:
-
Jacobs, J. W., Hall, E., Tahiri, T., et al. (2026). Direct donation of unvaccinated blood: A departure from evidence-based medicine associated with clinical harm, wasted resources, and surveillance gaps in a 2-year single-center series. transfusion. Toi: https://doi.org/10.1111/trf.70195. https://onlinelibrary.wiley.com/doi/10.1111/trf.70195