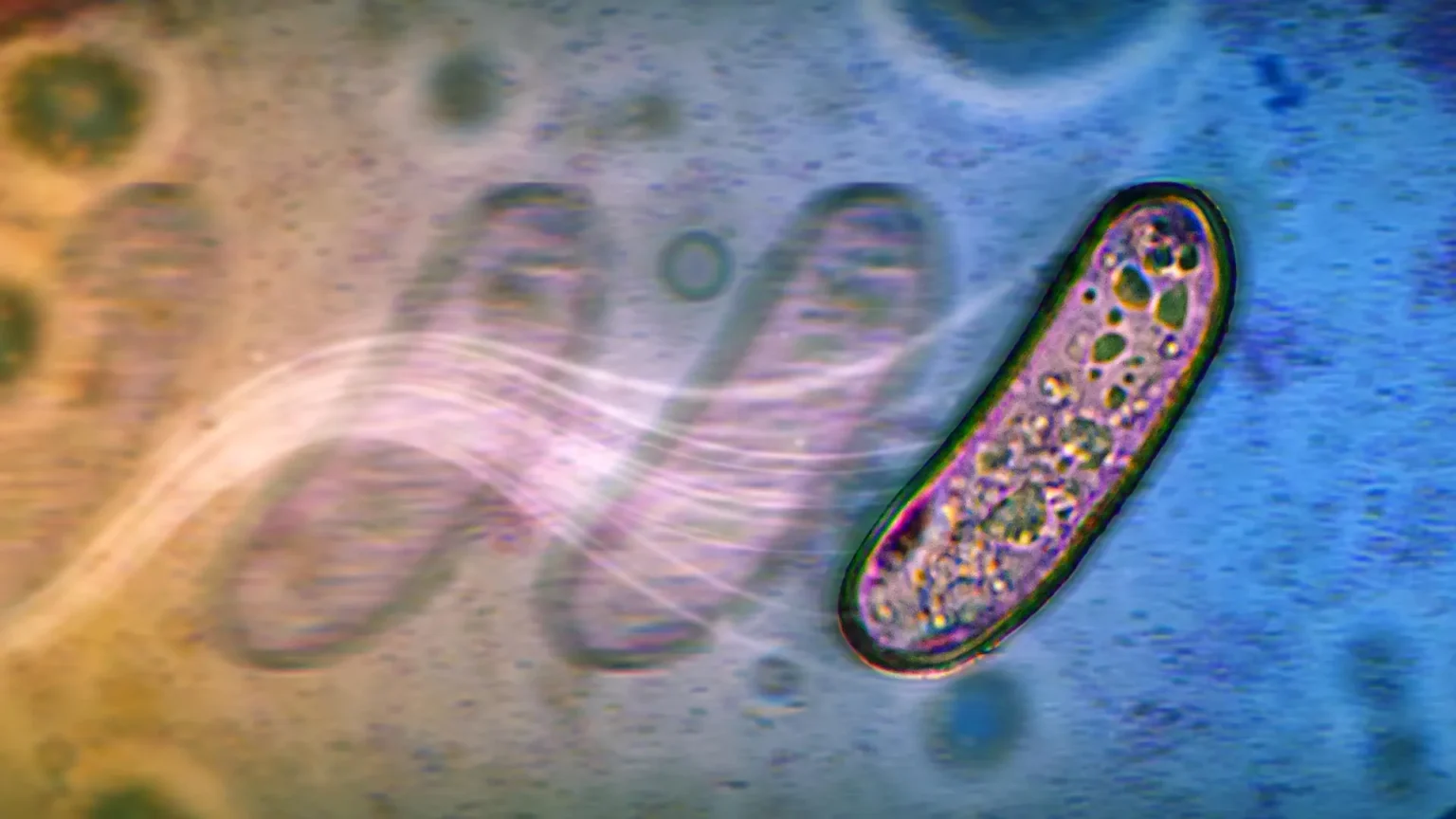
New research from Arizona State University shows that bacteria can move in unexpected ways even when normal propulsion systems fail. Bacteria normally move using flagella, long, thin whip-like structures that rotate to push the cell forward. New research has revealed that microorganisms can spread across surfaces even without these structures.
Movement is very important for bacteria. It allows them to gather in communities, explore new environments, and escape harmful situations. Knowing how bacteria move may help scientists develop better strategies to prevent infection.
In the first study, researcher Navish Wadhwa and his team found that salmonella and Escherichia coli Even when flagella are disabled, they are still able to move across moist surfaces. Bacteria generate movement through metabolism. As the sugar ferments, it creates small outward flows on the wet surface. These streams slowly push bacterial colonies outward, similar to leaves on a tree flowing along a narrow stream.
Researchers have dubbed this newly identified movement “swashing.” The discovery could help explain how disease-causing microorganisms colonize medical devices, wounds and food processing equipment. By understanding how bacterial metabolism causes this type of movement, scientists may be able to slow or stop it by changing environmental conditions such as pH or sugar levels.
“We were surprised by the ability of these bacteria to move across a surface without a functional flagellum. In fact, our collaborators originally designed this experiment as a ‘negative control,’ meaning we expected the cells to become immobile[once the flagellum was removed],” Wadhwa says. “But the bacteria moved on as if nothing were wrong, and we began a multi-year quest to understand how they do it.
“This shows that even when we think we have something figured out, surprises are often waiting just below the surface, or in this case above the surface.”
Wadhwa is a researcher in the Biodesign Center for Evolutionary Mechanisms and an assistant professor in the ASU Department of Physics. This research bacteriology journal It was selected as an Editor’s Pick, highlighting its importance.
swashing with sugar
The swashing effect begins when bacteria consume fermentable sugars such as glucose, maltose, and xylose. During fermentation, microorganisms release acidic byproducts such as acetate and formate. These compounds draw water from surrounding surfaces toward the colony, creating small currents that push the cells outward.
This movement requires fermentable sugars. Without these, the bacteria cannot create the fluid flow necessary for swashing. Therefore, an internal environment rich in sugars, such as mucus, can make it easier for harmful bacteria to spread and cause infections.
The scientists also tested what happens when they add surfactants, detergent-like molecules, to the colonies. These compounds completely stopped the swash. However, the same chemicals did not prevent swarming, another type of bacterial movement driven by flagella that allows microorganisms to spread rapidly across wet surfaces. This difference suggests that the two behaviors rely on separate physical mechanisms. It also suggests that surfactants may one day be used to control bacterial movement, depending on whether the microbes swarm or swarm.
The discovery that bacteria can colonize surfaces even when the normal swimming mechanism fails has important health implications. Some microorganisms can be spread throughout medical catheters, implants, or hospital equipment through cleaning. Blocking flagella alone may not prevent its spread. Instead, treatments may need to target the metabolic processes that cause fluid flow.
Escherichia coli and Salmonella are both well-known causes of food poisoning. Recognizing that these bacteria can spread through passive fluid flow may help improve sanitation strategies in food processing facilities. Because swashing relies on fermentation and acidic byproducts, changing factors such as surface pH and sugar levels can limit bacterial growth. The study found that even small changes in acidity can affect bacterial migration.
A similar condition may exist within the human body. Moist environments such as intestinal mucus, wound fluids, and the urinary tract provide surfaces on which bacteria can spread by being washed away, even when flagella are not functioning effectively.
Molecular gear system that supports bacterial movement
The second study looked at another group of microorganisms called Flavobacteria. Unlike Escherichia colithese bacteria do not swim. Instead, they use a specialized machinery known as the type 9 secretion system (T9SS) to move along the environment and surfaces of their hosts. This system powers a molecular conveyor belt that moves along the surface of the cell.
Under normal conditions, T9SS allows flavobacteria to glide across surfaces. The mechanism works by running an adhesive-coated belt around the outside of the cell, pulling the bacteria forward in a motion similar to a microscopic snowmobile.
The researchers discovered that a protein in this system called GldJ acts as a kind of gear shifter that controls the direction of the motor. When a small portion of GldJ is removed, the motor reverses its rotation from counterclockwise to clockwise. This change changes the direction of bacterial movement.
This study details this molecular gearing and shows how it allows bacteria to adjust their movements in response to complex environments. This ability may provide an evolutionary advantage by allowing microorganisms to move across surfaces more effectively.
Implications for human health and microbiome research
The T9SS system not only affects bacterial movement; They can also impact human health in different ways, depending on the microbial communities involved.
In the oral microbiome, bacteria containing the T9SS system have been associated with periodontal disease. The proteins they release can cause inflammation in the mouth and can also lead to conditions such as heart disease and Alzheimer’s disease.
In contrast, T9SS activity in the gut microbiota may be beneficial. Proteins secreted through this system can prevent antibody degradation, which may strengthen immune defense and improve the effectiveness of oral vaccines.
Understanding how this molecular gearbox works could help researchers develop ways to stop bacteria from forming biofilms, or slimy communities, that cause infections and contaminate medical devices. At the same time, scientists may be able to harness these mechanisms to support beneficial microbes and design targeted microbiome treatments.
“We are very excited to have discovered an extraordinary dual-role nanogear system that integrates feedback mechanisms, revealing a controllable biological snowmobile and showing how bacteria precisely adjust their motility and secretion in dynamic environments,” says Shrivastava. “Building on this breakthrough, we now aim to determine the high-resolution structure of this remarkable molecular conveyor and visualize with atomic precision how its moving parts work together, transmit forces, and respond to mechanical feedback. Unraveling this complex design will not only deepen our understanding of microbial evolution, but will also inspire the development of next-generation bioengineered nanomachines and therapeutic technologies.”
Shrivastava is a researcher in the Biodesign Center for Basic and Applied Microbiomics, the Biodesign Center for Mechanisms of Evolution, and an assistant professor in the ASU College of Life Sciences. The discovery will be published in a magazine mBio.
Multiple strategies help spread bacteria
At first glance, the two discoveries of fluid surfing and molecular gearshifting seem completely different. But both highlight how bacteria have evolved a variety of unexpected strategies to move and spread. The more movement options a microorganism has, the more difficult it is to control.
These findings also suggest that new approaches may be needed to combat bacterial infections. Many traditional strategies focus on disabling flagella. But these studies show that bacteria can continue to spread even in their absence.
This study points out the importance of controlling the environment in which bacteria live. Factors such as sugar availability, pH levels, and surface chemistry can play a major role in limiting bacterial migration. Interfering with molecular systems like the T9SS gearbox can prevent bacterial movement and the release of harmful proteins that cause disease.